Table of Contents
Graham Blackbeard,
Founder and Managing Director
Message from Graham
Welcome to the July issue of “Southern Implants In Focus”
Each time I look at social media, I am inspired (and overwhelmed) by the magnificent treatments that I see are being given to patients, whilst making optimal use of our products. My sincere appreciation of the support we have received throughout this pandemic.
Not only at Head Office, but in many countries, we are adding staff members to our teams. I am sure you will join me in welcoming them to the Southern Family.
We had been preparing for the new MDR transfer audit for eight months, and we hosted the notified body for the MDR transfer audit during March.
We chose to be one of the first firms in the world to be audited in terms of the new European MDR. The audits have gone well and we are on our way to become one of the first Medical Device companies to be MDR certified.
In February, Southern attended the Academy of Osseointegration (AO) virtual meeting. Alex Stuart-Smith, one of our dedicated engineers, won 2nd prize for her presentation regarding our ASC (Angulated Screw Channel) system which she is developing. Read more on her presentation in this newsletter. ASCs have gained a reputation of being associated with “screw loosening”, resulting from a decreased preload in the retaining screw due to the angulation. In this study, Alex proposes a new screw channel utilising high strength materials and optimum dimensions, generating significantly higher preload than current ASCs.
Predictable, accelerated implant treatments are more important than ever before. Recent research reveals that adapting clinical practice with innovative treatment solutions provided significant benefits, that not only impact positively on patient satisfaction, but also enable clinicians to provide more treatments. Read more on this research by Drs Morris, Steinberg and Drago here.
We’re excited to be rolling out SIREAL, the Southern Implants guided surgery solution, this year. Our aim is to offer a truly universal system, providing the complete computer-assisted dental implant planning and placement solution for Southern Implants’ tapered implants. Read more about it in this article by Stuart Blackbeard.
Until next time, thank you for being part of the Southern family.
SIREAL™ Guided Surgery
By Stuart Blackbeard
Overview
When you think of guided implant surgery, what you should be thinking is; I have done all the legwork and planning beforehand, therefore this should make preforming the procedure simple and efficient. However, a lot of the time, this is not the case! A multitude of planning software, 3D printers and everything else in-between, results in your first hurdle.
The second focus is the associated instrumentation, and this is where it gets really fun; dedicated guided surgery kits for specific implant ranges with a magnitude of drills, spoons, spacers and placement tools depending on what you are trying to achieve. The costs associated with this already start to raise many eyebrows, along with being restricted to the plan with little margin for adapting to bone quality and the like. Following this is preforming the actual procedure, with complicated drilling sequence recipes, spoons popping in and out and different drills for different length and size implant requirements.
In my mind, this all sounds like a headache. Something that should be quick and easy has now turned into a nightmare of accumulating instrumentation, increased costs and lack of simplicity.
Surely there is a way to use what we already know about planning and printing and combining this with standard drills and surgical instruments to conduct guided implant surgery. The question I have always asked is “Why does one need a special separate surgical kit, there must be a way of making standard systems guided?”. Well, this is the question that we, at Southern Implants, have been thinking about for a couple of years, especially when considering the fact that Southern Implants has five different implant connection systems. We cannot simply exclude some clinicians because of the connection system they prefer to use. Therefore, a universal guided surgery solution was the only way to go.
There was definitely no ‘quick fix’ in the endeavour to finding our final solution. Thankfully, this process was made significantly easier, due to our loyal global customer base, always willing to provide important insight and support throughout the system’s development.
In 2020, the SIREAL™ system was finally born!
SIREAL™ is the Southern Implants guided surgery solution, providing the complete computer-assisted dental implant planning and placement solution for Southern Implants’ tapered implants. This is by virtual prosthesis and on-screen design of surgical guides, allowing prosthetically driven implant placements. The SIREAL system supports the full drilling protocols of all our Dental Implant Ranges: DC (Deep Conical), External Hex, TRI-NEX®, Inverta®, IT (Internal Octagon), PROVATA® and Internal Hex.
SIREAL™ Guided Surgery aims to provide a truly universal system. To develop a universal system, our system needs be compatible with the majority of the planning software on the market, as this allows customers the freedom of personal preference as to which software they use. The use of multiple guided drills, spoons and spacers also need to go! This approach is cumbersome, creates confusion and can result in inaccuracies during drilling and placement.
Planning Software
Starting with the planning of a case and the various software options available: they all make use of 3D imaging to plan the positioning of the implant. Based on this, the surgical guide is then designed and printed. The Southern Implants implant library is currently included in 25 planning software packages with many more on the way.
The Beauty of the SIREAL™ system
For many so called “universal systems”, preparing a site-specific osteotomy is complicated and often only works for parallel-walled implants or when creating a stepped osteotomy. However, Southern customers like to place our tapered implants, therefore our site-specific drills need to be utilised for optimum implant placement.
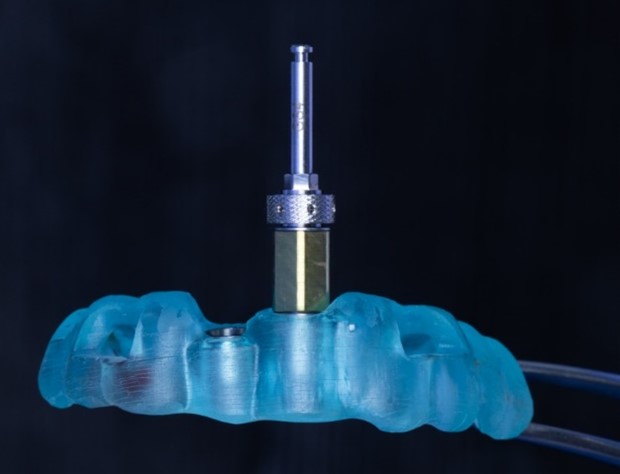
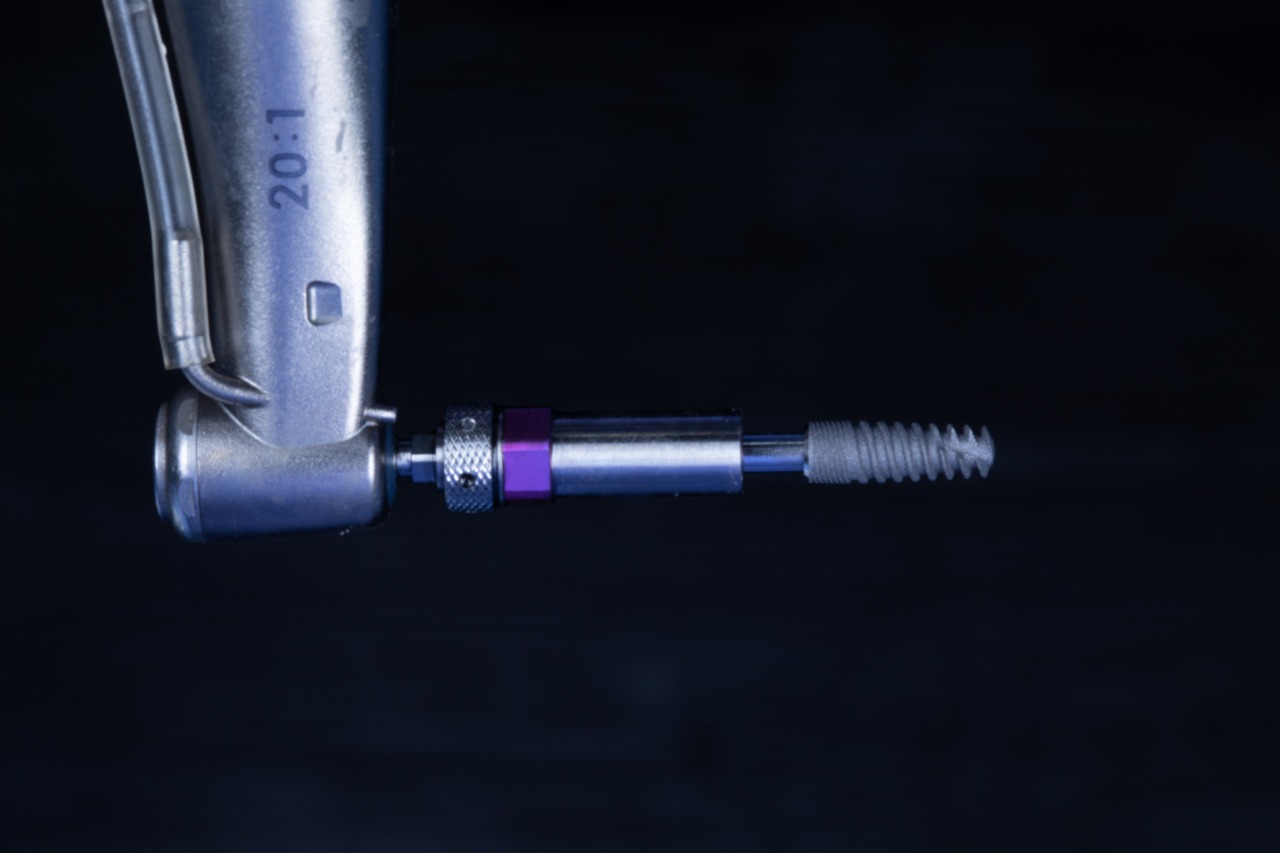
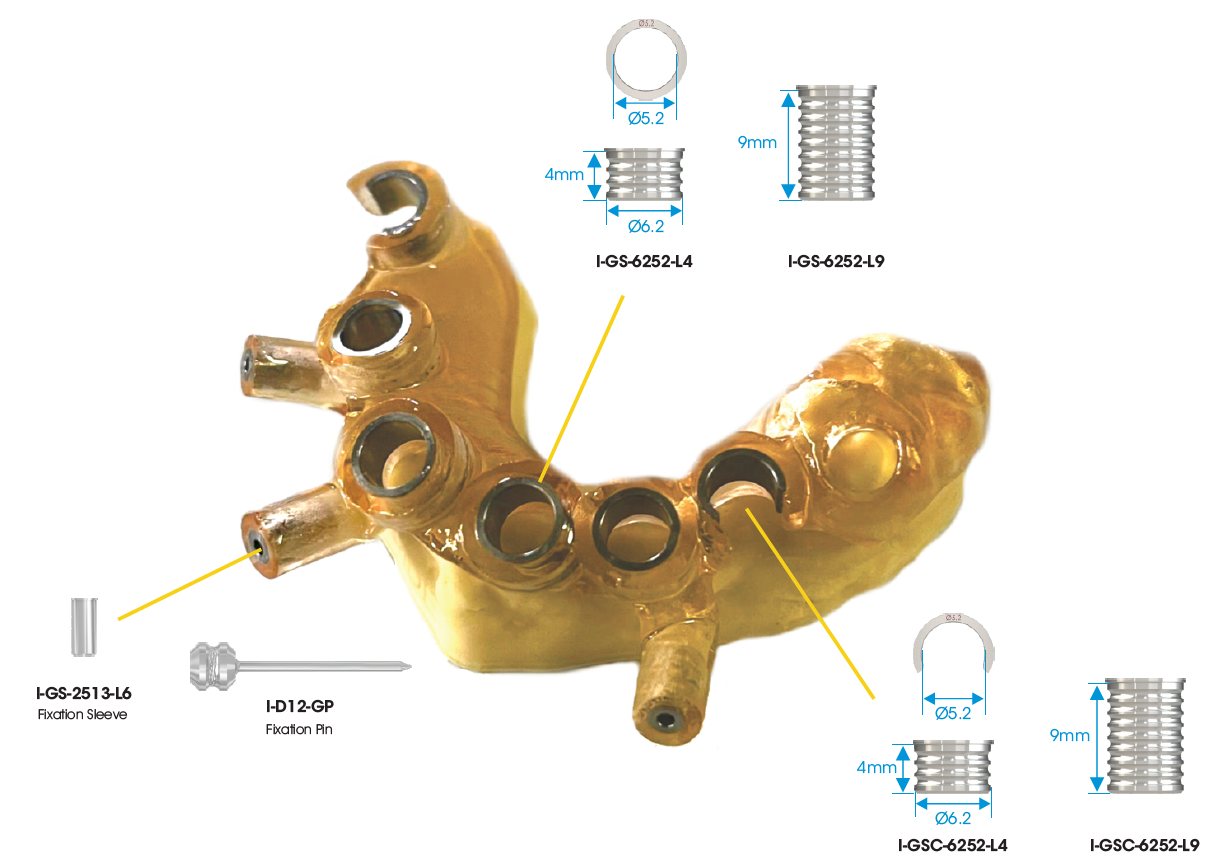
The universal guided surgery tool from Southern Implants, affectionately known as “The Cannon”, is the key element of the SIREAL Guided Surgery system. This tool allows clinicians to utilise their standard Southern Implants drill kits and converts your tapered drills into guided surgery drills. This unique system extends to other tools in the surgery as well. The Cannon features a “twist lock” mechanism which locks the latch of tools tightly in place. The W&H hex allows for Southern torque wrench and W&H compatible handpieces to engage The Cannon and apply high torques.

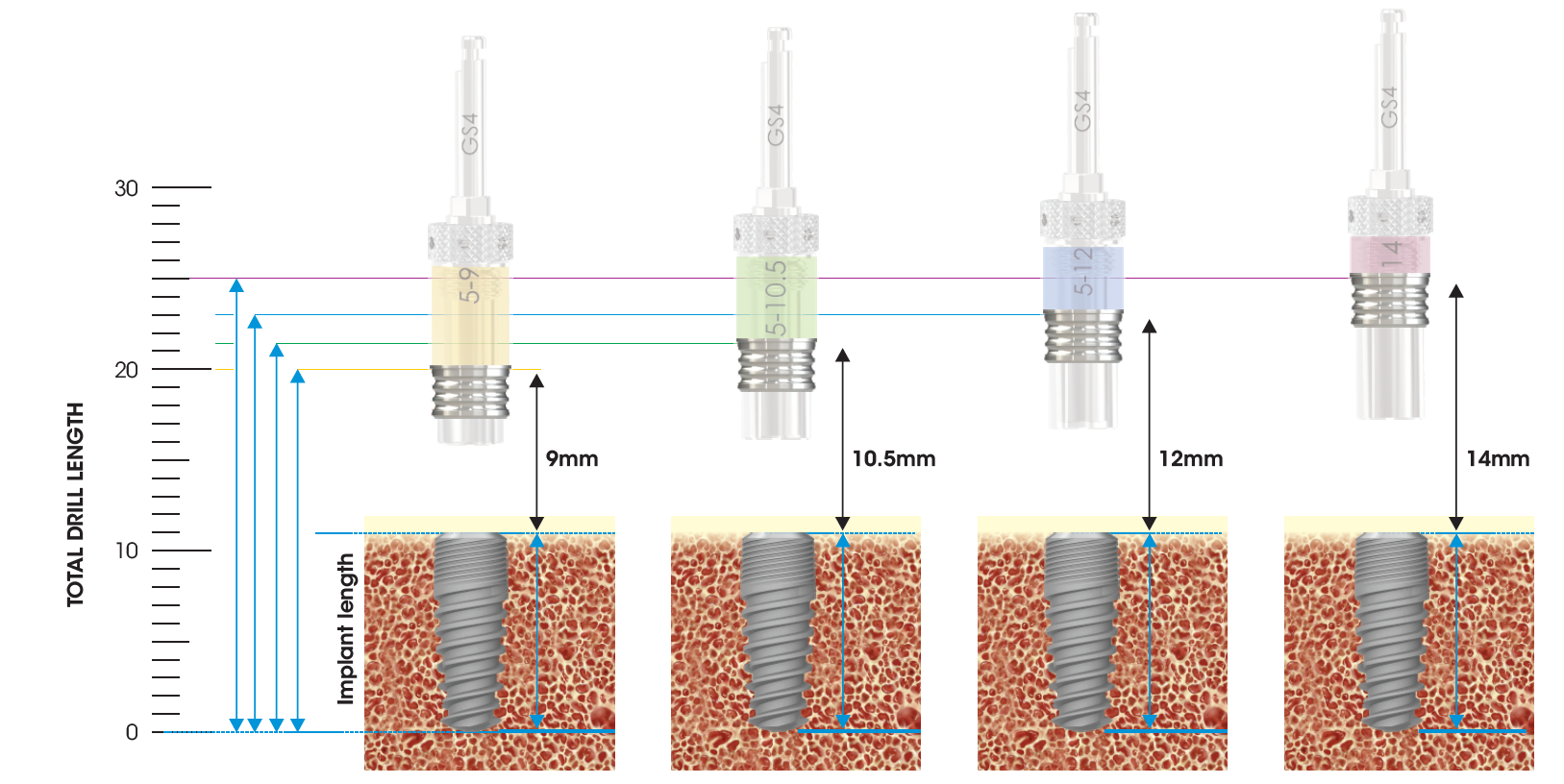
The Cannon is designed with offset sleeve threads to attach the different Offset Sleeves. These offset sleeves determine the hole/sleeve diameter and the guide positional offsets. Two diameters are available; Standard (Ø5.1) and Narrow (Ø4.1). The SIREAL™ offsets are 9mm, 10.5mm, 12mm and 14mm in length. The combination of the standard & narrow diameter offsets sleeves provides versatility in planning to accommodate for a patient’s anatomical constraints (vertical opening, adjacent teeth etc.) which could interfere with the guide sleeve.
Surgical Guide Options
Depending on a clinician’s personal preference, there are three guided options available:
- Pilot Drill Guided – a pilot guide solution for the initial pilot drill only, with the osteotomy finished freehand.
- Partially Guided (full drill protocol) – full drilling protocol through the surgical guide.
- Fully Guided – All drilling and implant placement, to planned depth and angulation, through the surgical guide (compatible with selected implants only).
Therefore, depending on the type of guided surgery preferred by the clinician, drill guide sleeves for pilot drill guides as well as partially or fully guided sleeves are available.
The SIREAL™ guided system offers two types of sleeve: a closed sleeve and the C-sleeve. The C-sleeve is particularly useful when placing Co-Axis® implants and will also assist the user to remove the fixture mount or placement tool when placing the implant through the guide. The C-sleeve is also extremely useful in the posterior region, where vertical opening is a challenge. The Cannon and drill can then be brought in from the side, allowing the practitioner to gain additional space of the offset distance. The C-sleeve also allows for irrigation at the osteotomy site while drilling.
The surgical guide type (bone-, tissue- or tooth-supported) is dependent on clinician preference, patient anatomy and the available planning software. Southern Implants also has fixation pins available to stabilise the surgical guide if necessary.
Southern Implants guided surgery system is simple, accurate and cost effective. Forget expensive guided surgery kits only for a specific implant range, with a magnitude of drills, spoons, spacers and placement tools depending on what you are trying to achieve. With SIREAL™ as tool at your disposal, Southern Implants has once again found an innovative solution to optimise and support your practice!
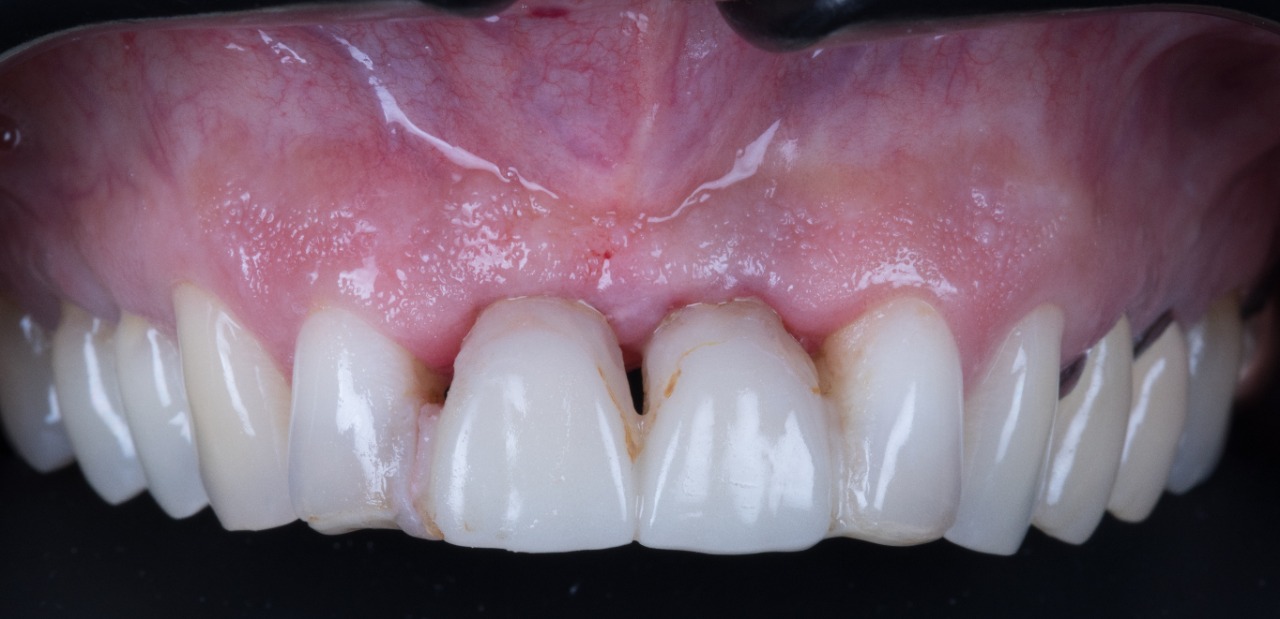
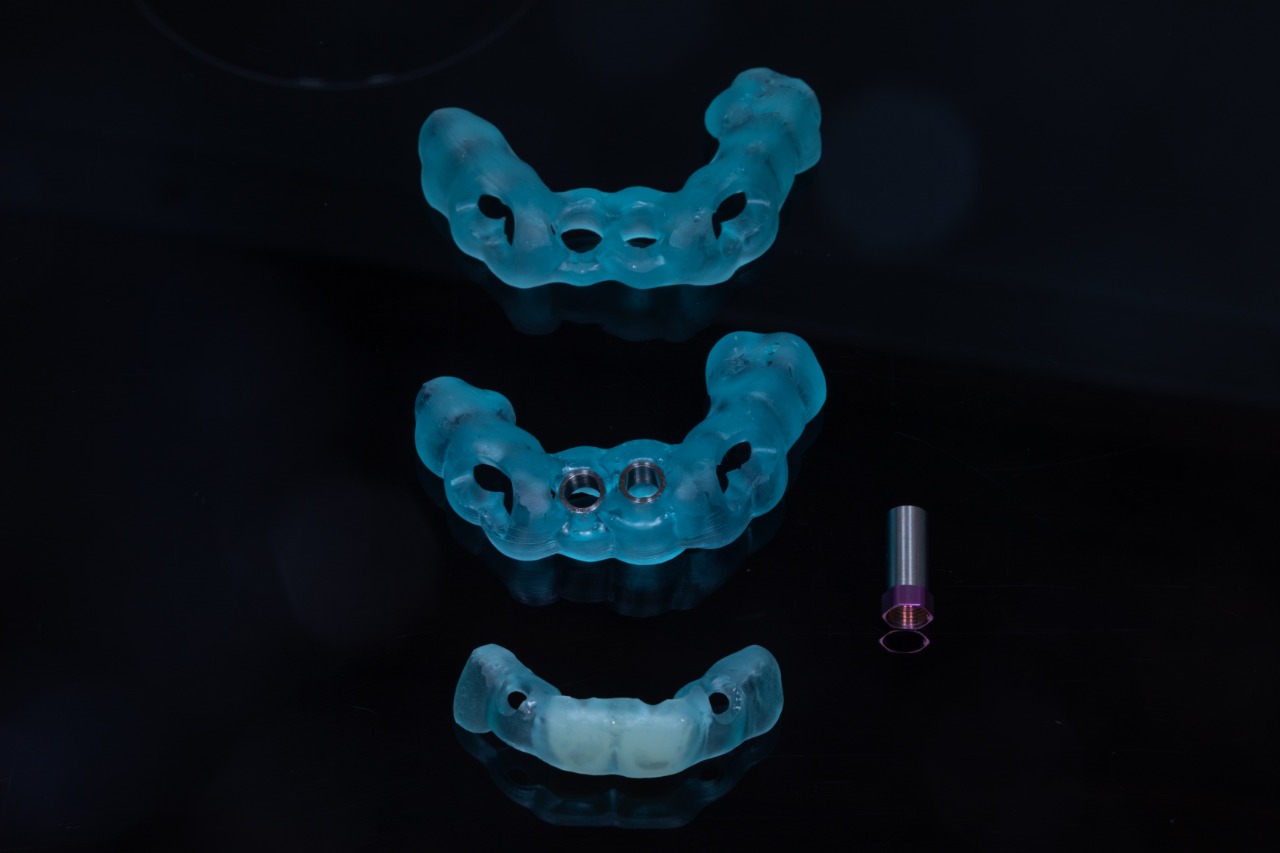
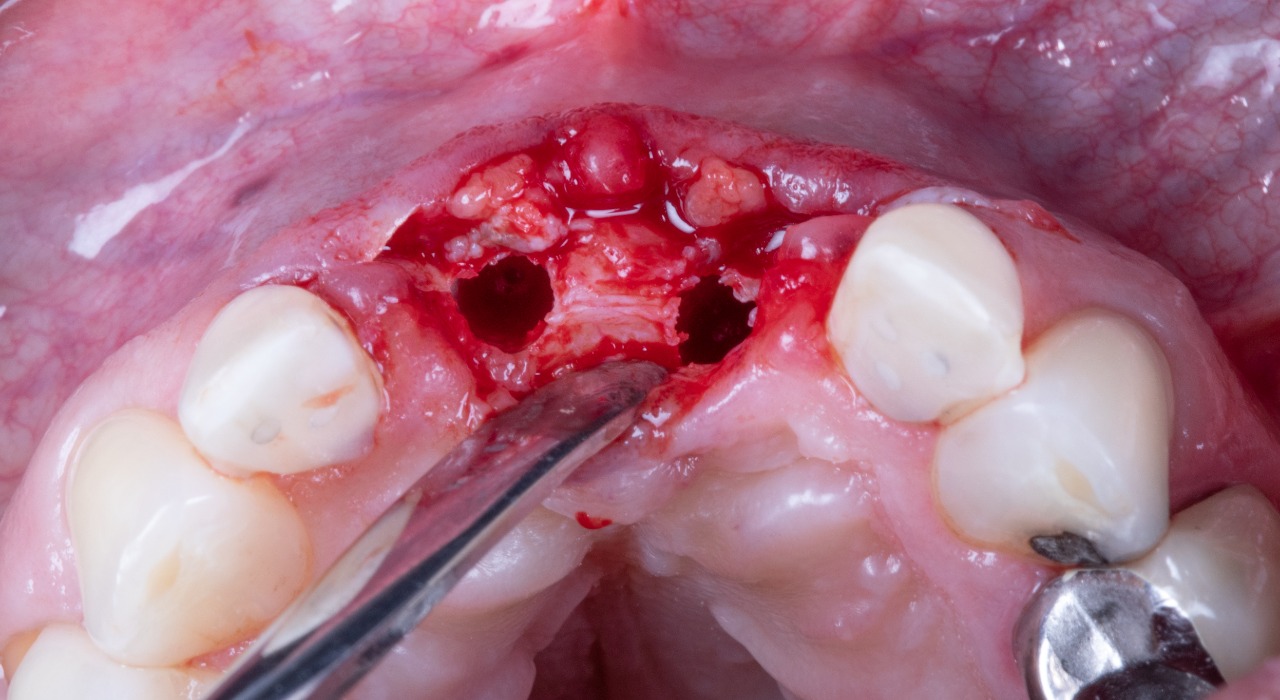
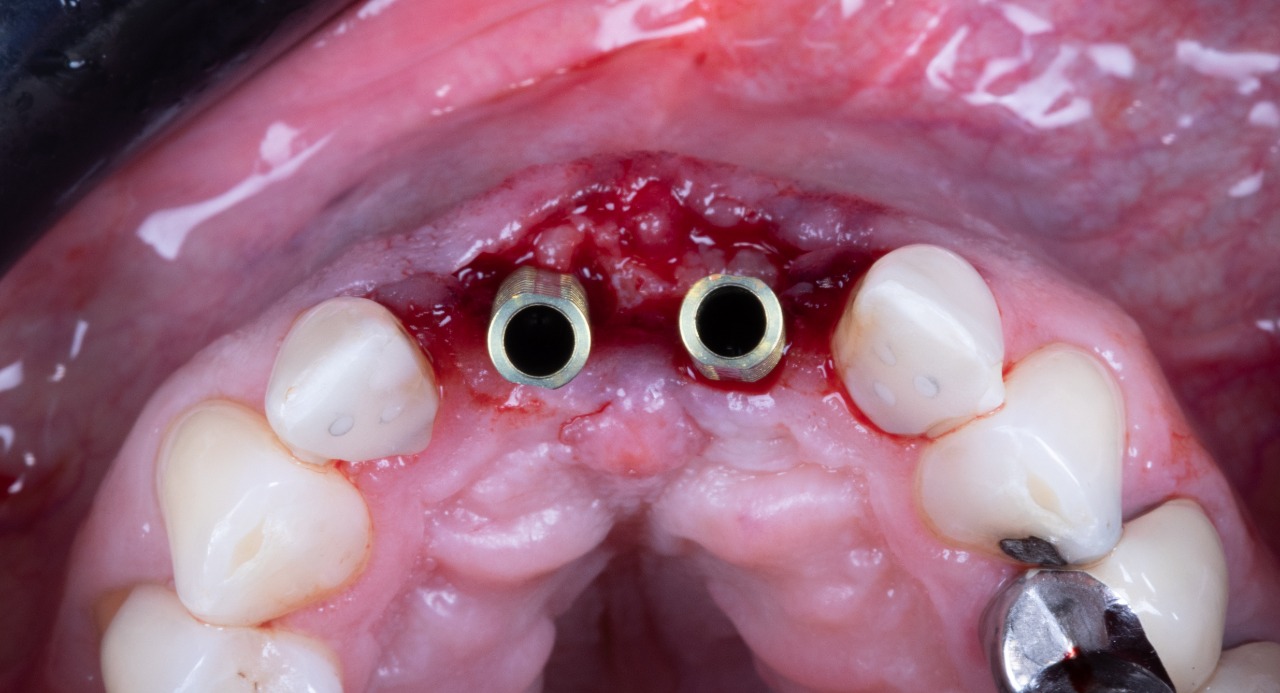
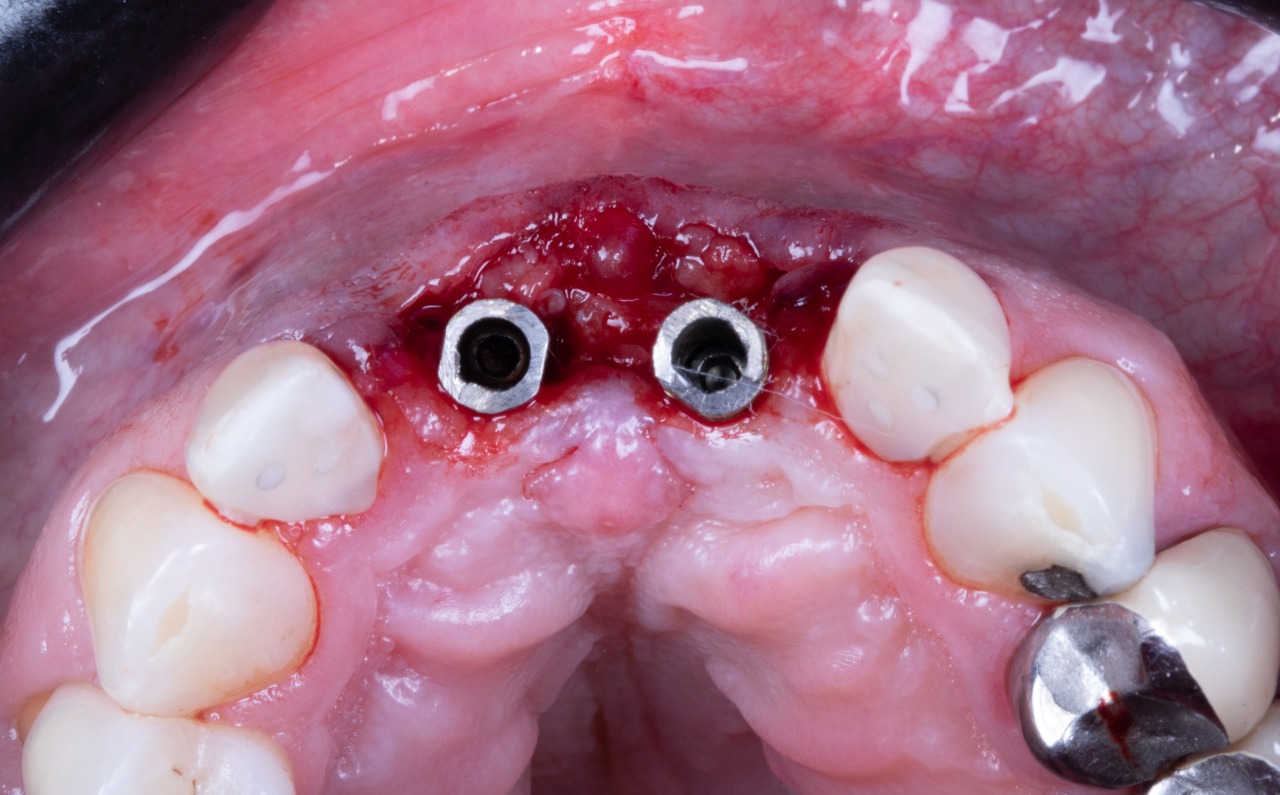
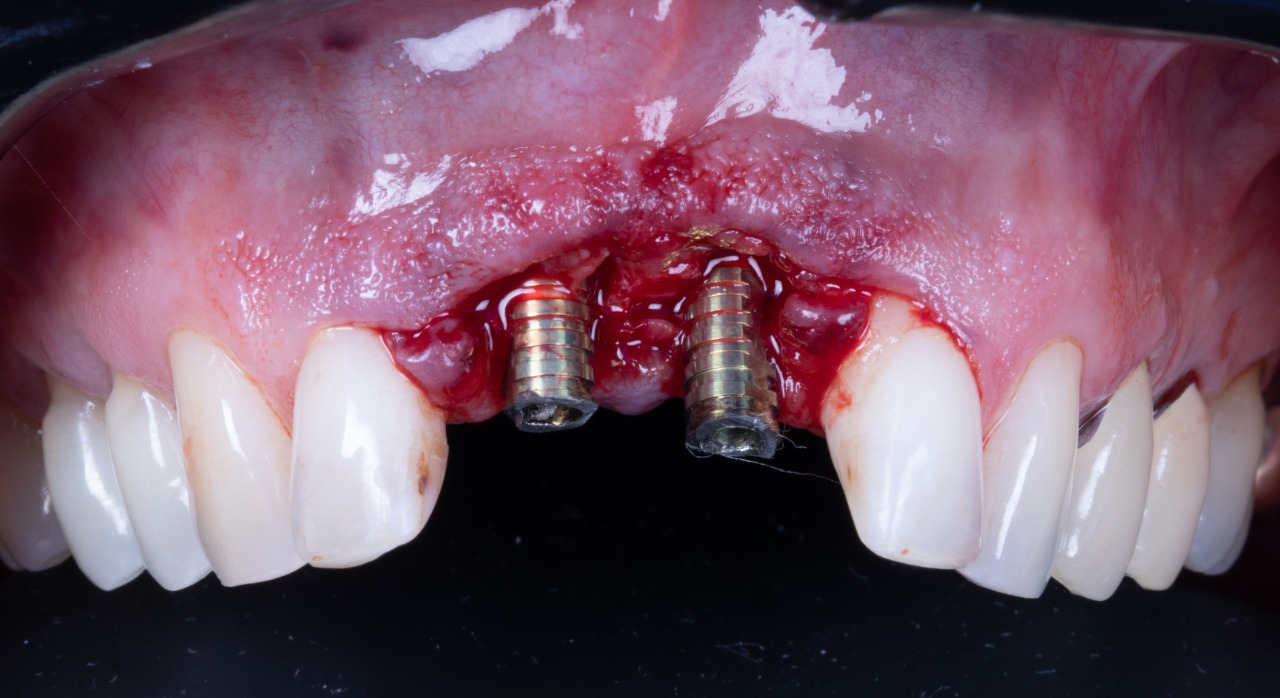
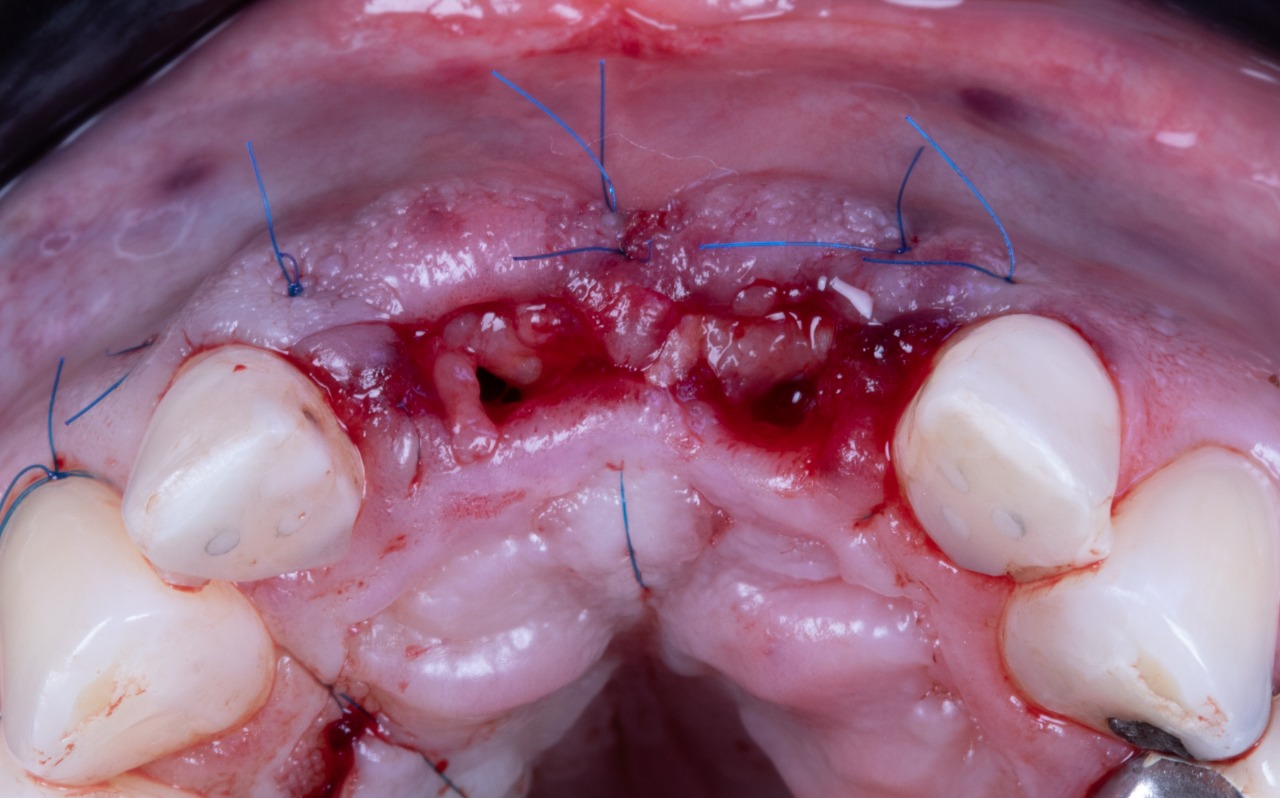
Missing Upper Anterior Centrals
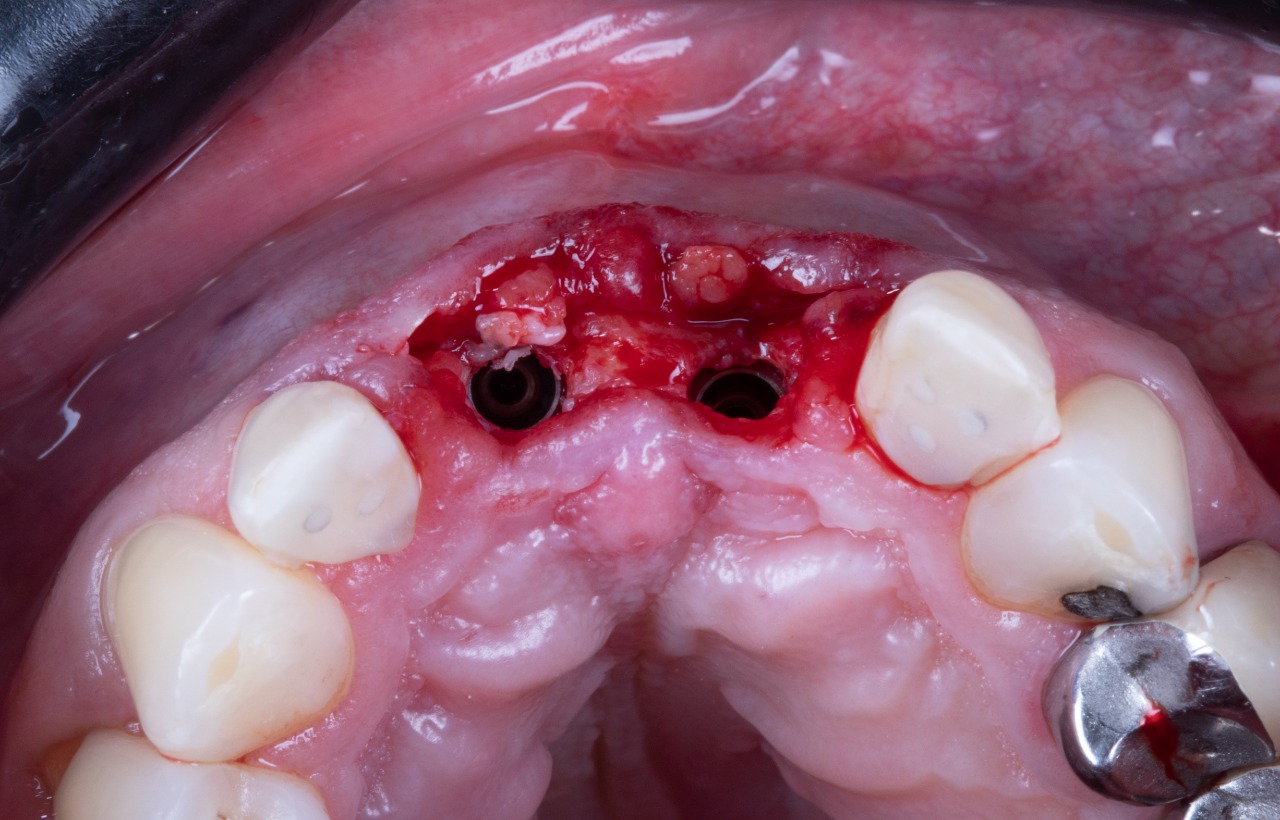
Missing Upper Anterior Centrals
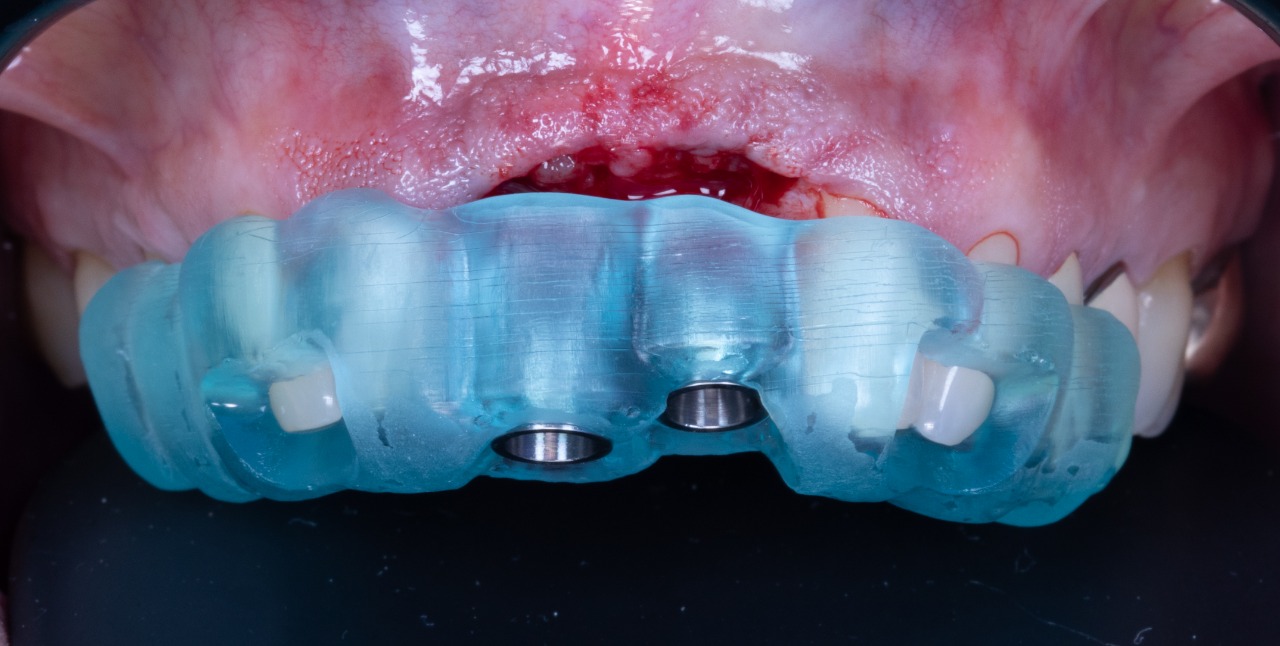
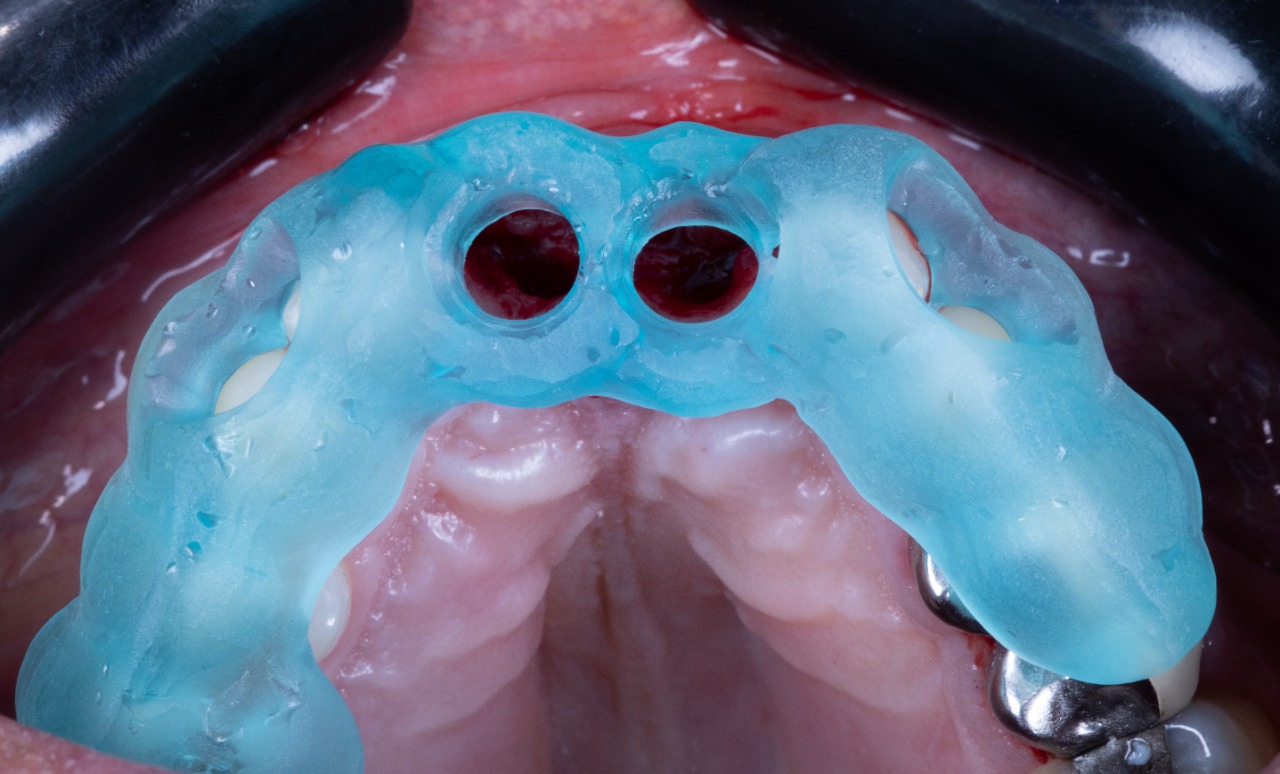
Implant placement guide, Drilling guided and Temporary Crowns
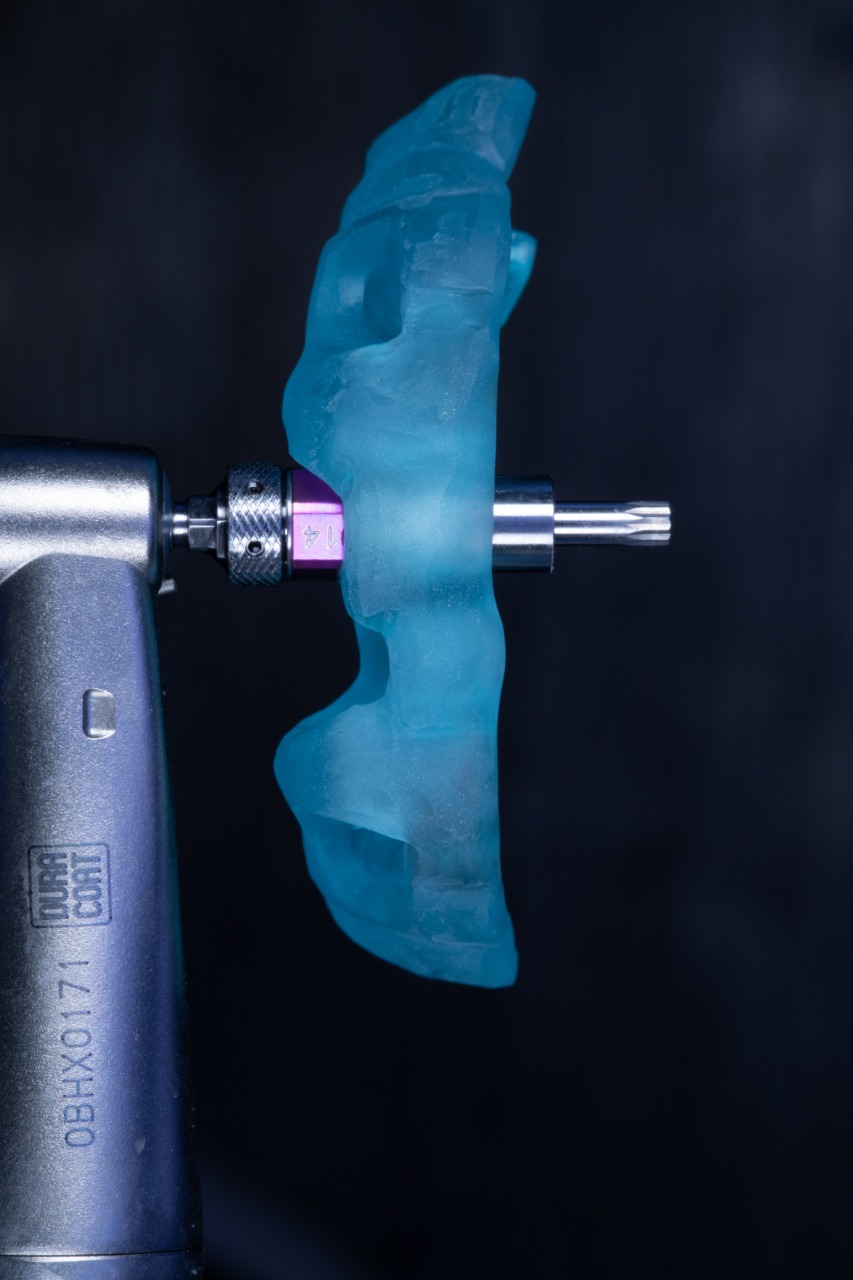
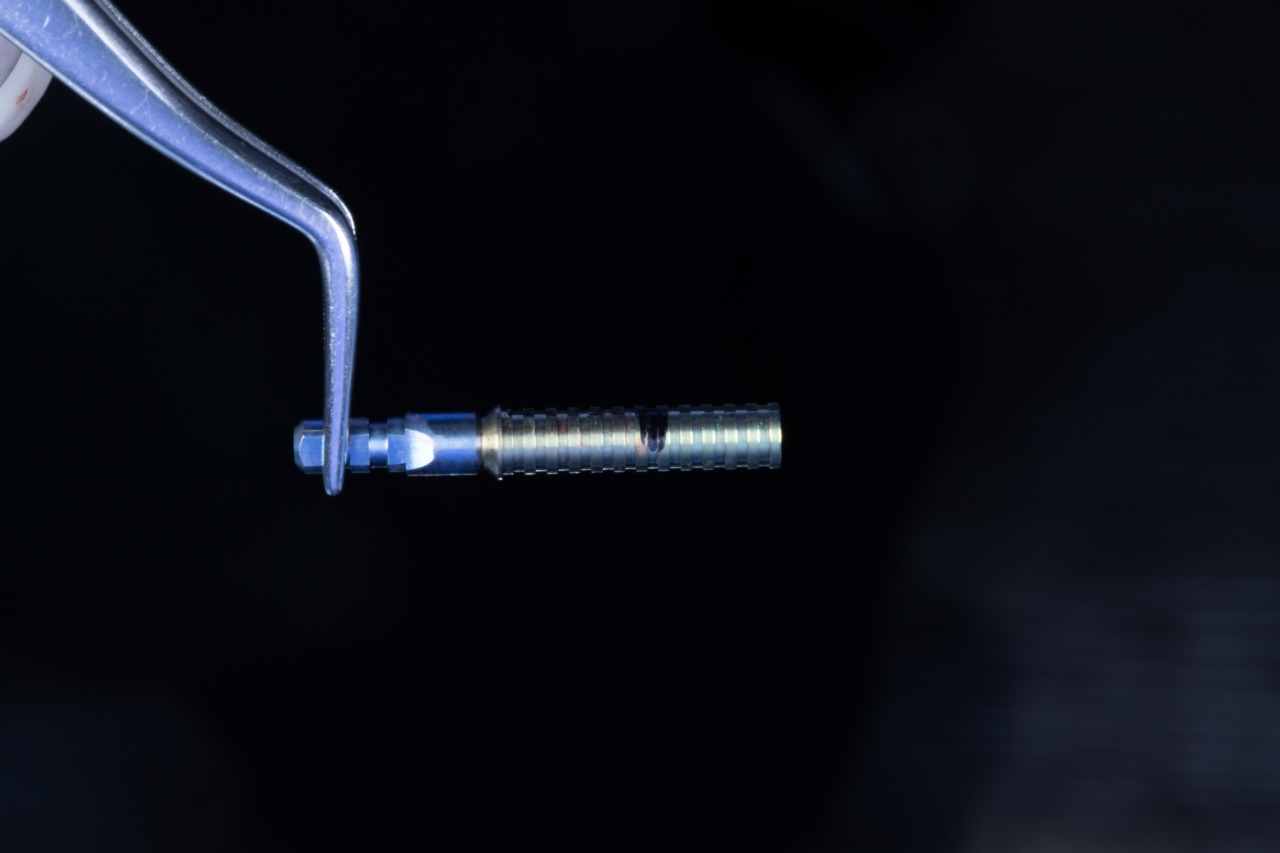
SIREAL Cannon with Offset Sleeve engaging the Drilling Guide
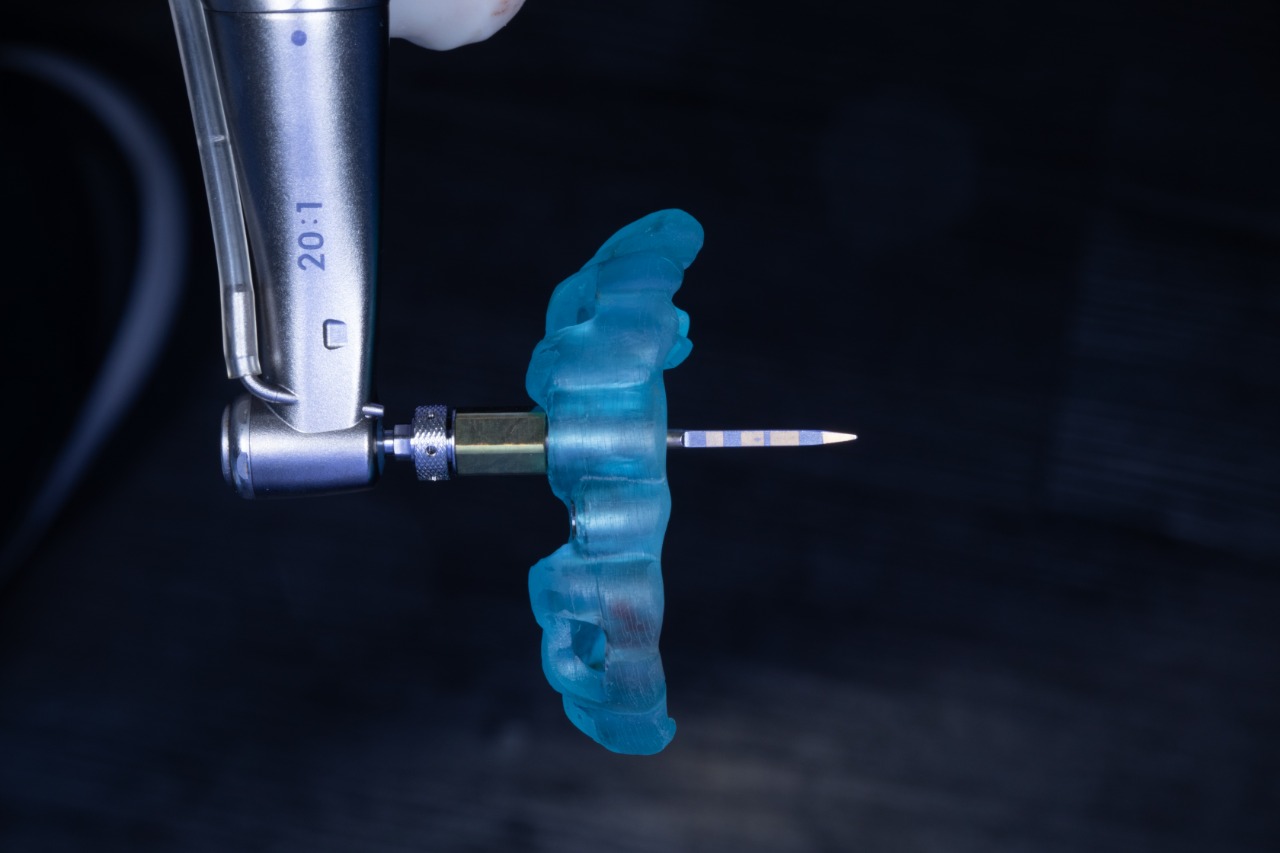
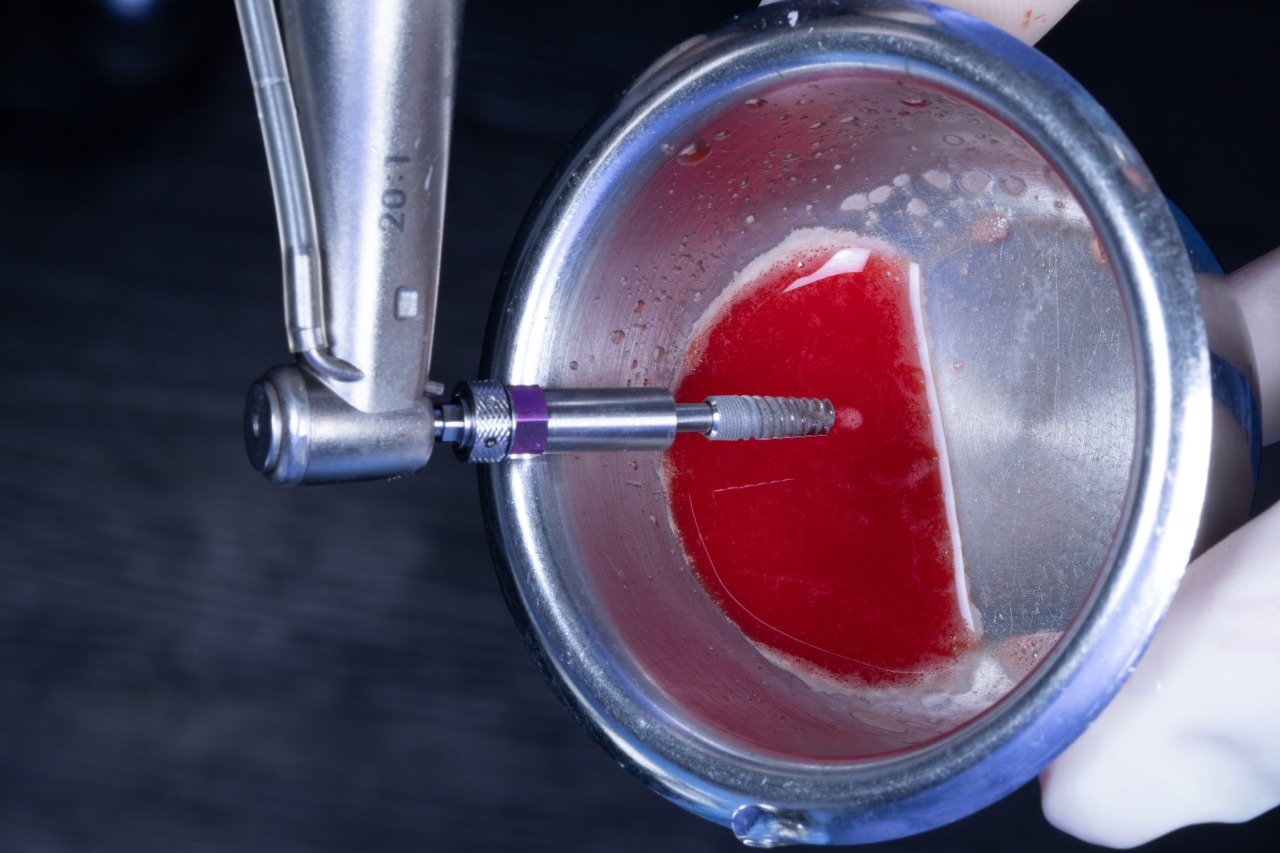
Initial drill in the Drilling Guide
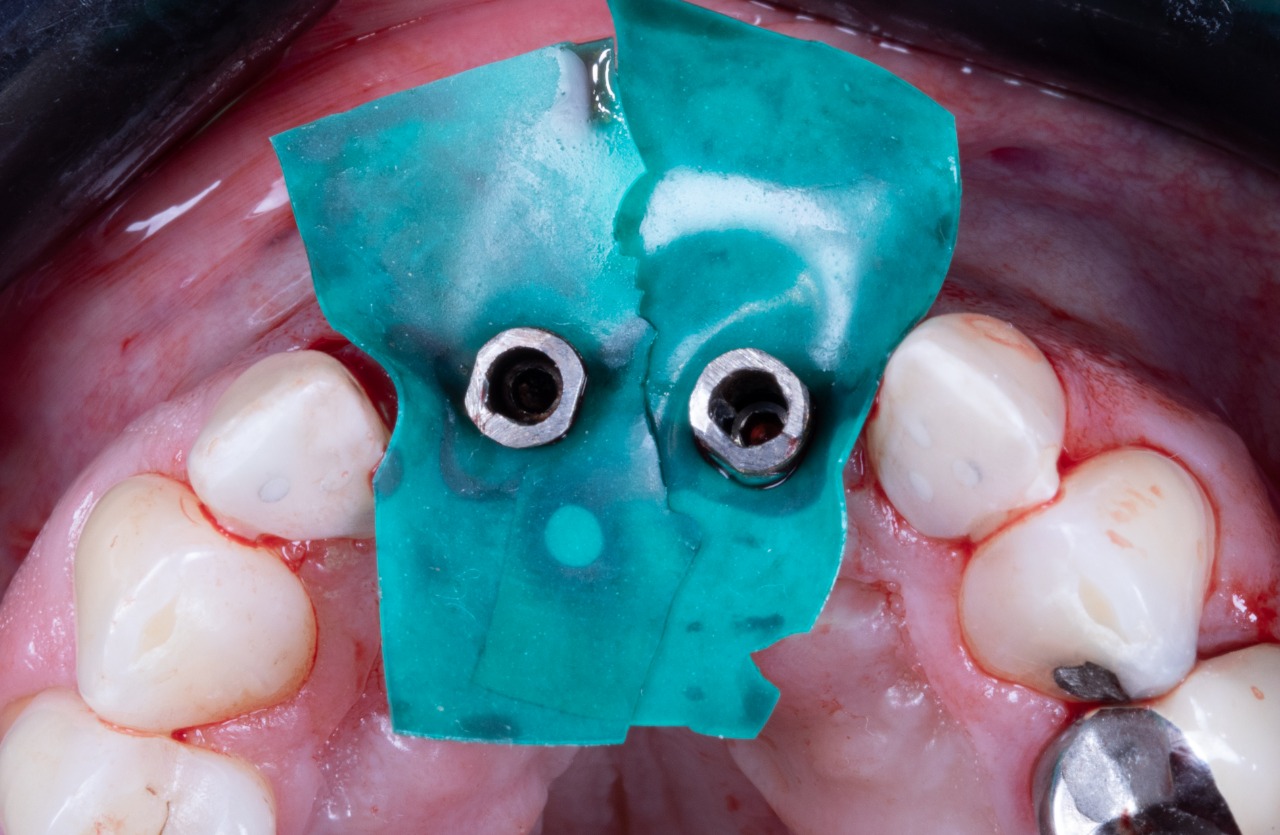
Drill Guide Seated (Bottom View)
Drill Guide Seated (Front View)
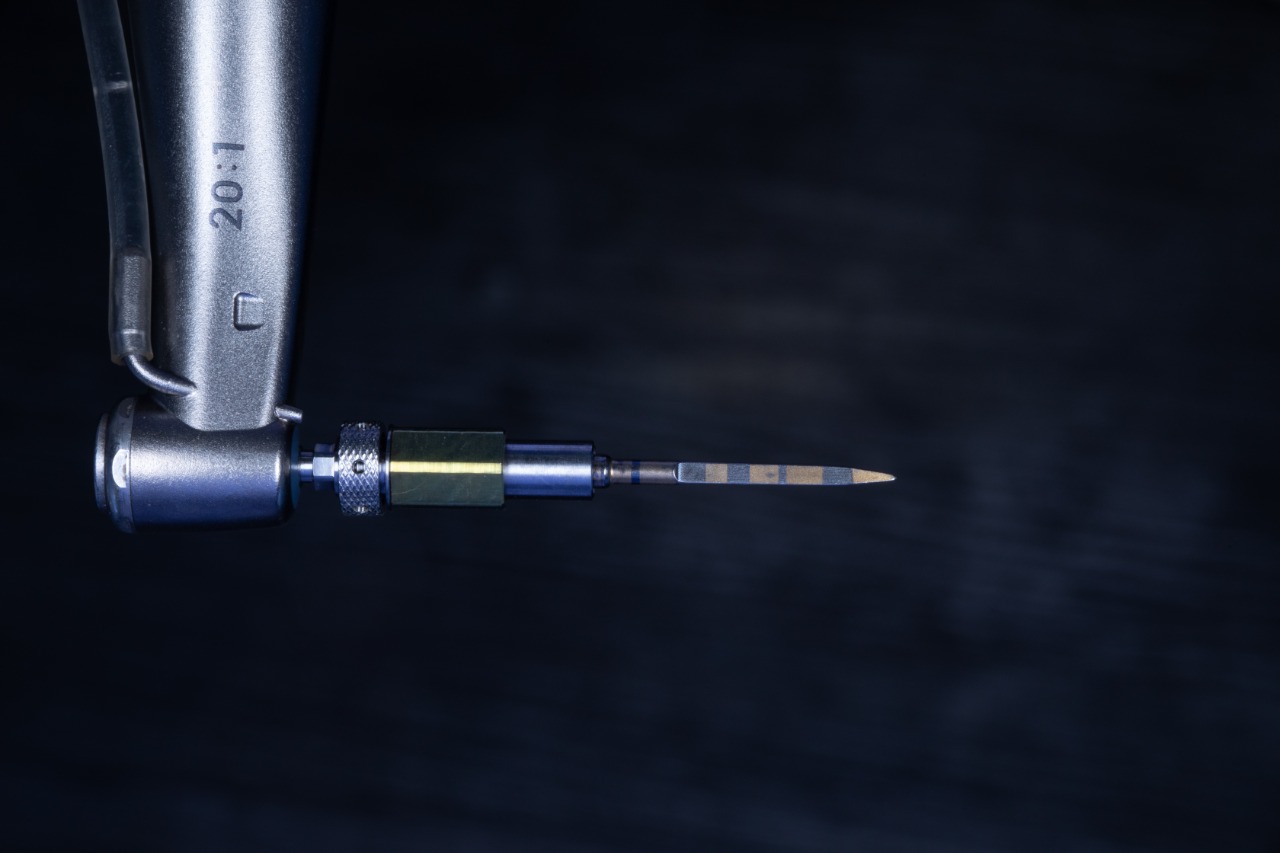
Initial drill
Direction Indicators position verification (Bottom View)
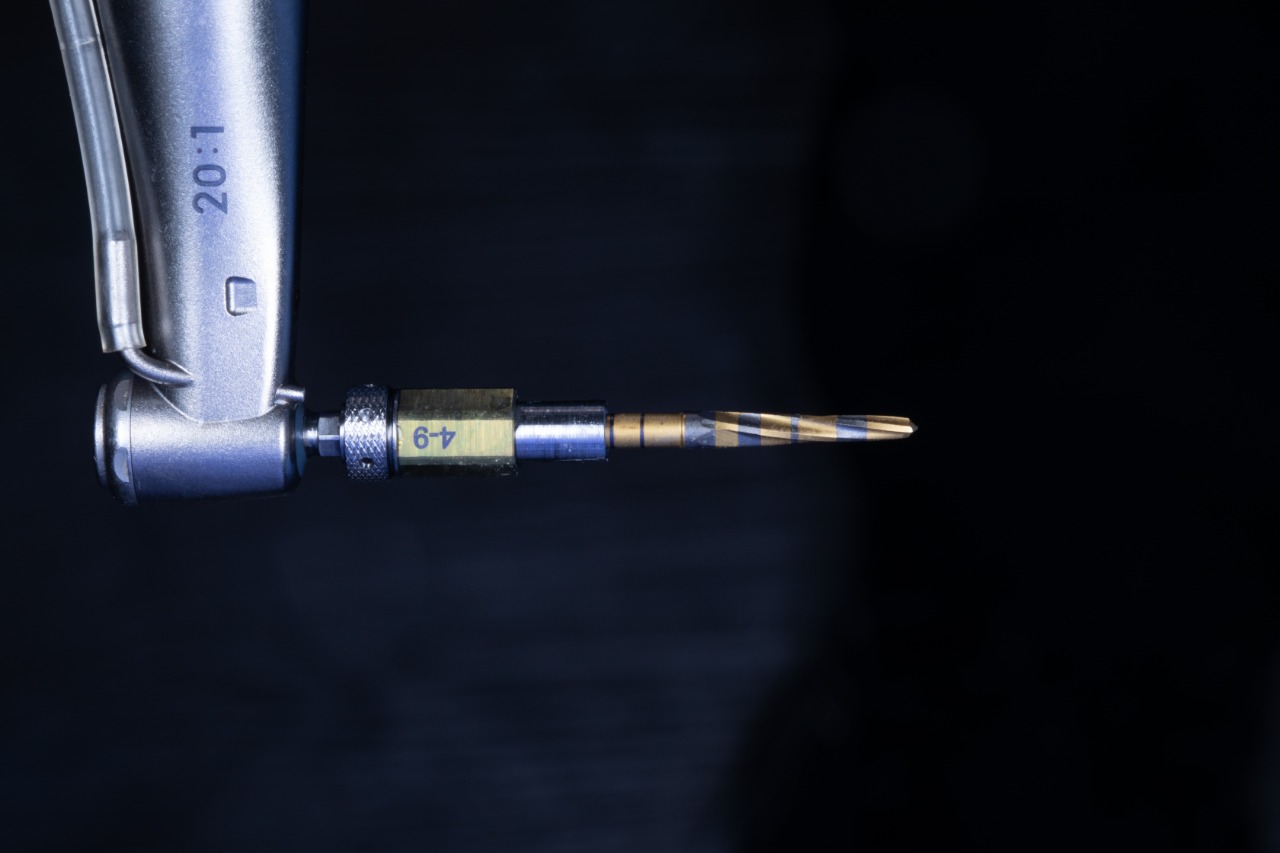
Final drill
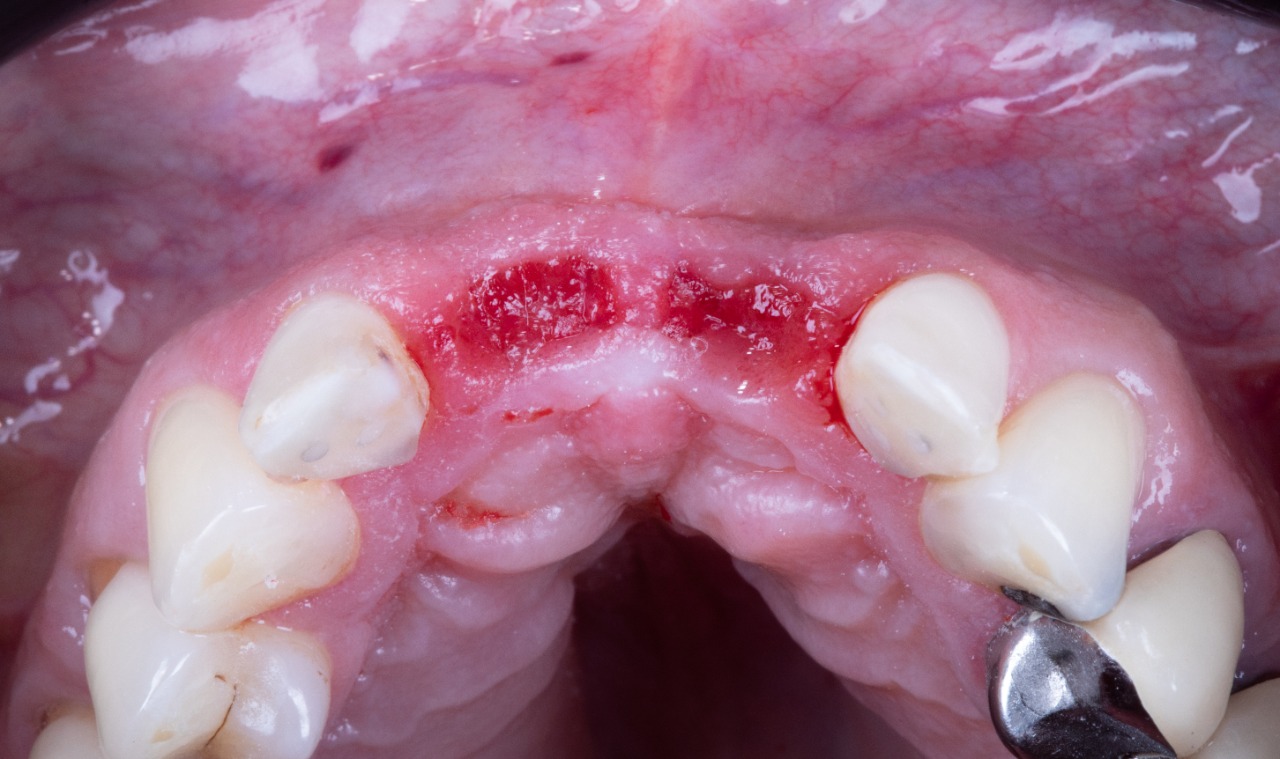
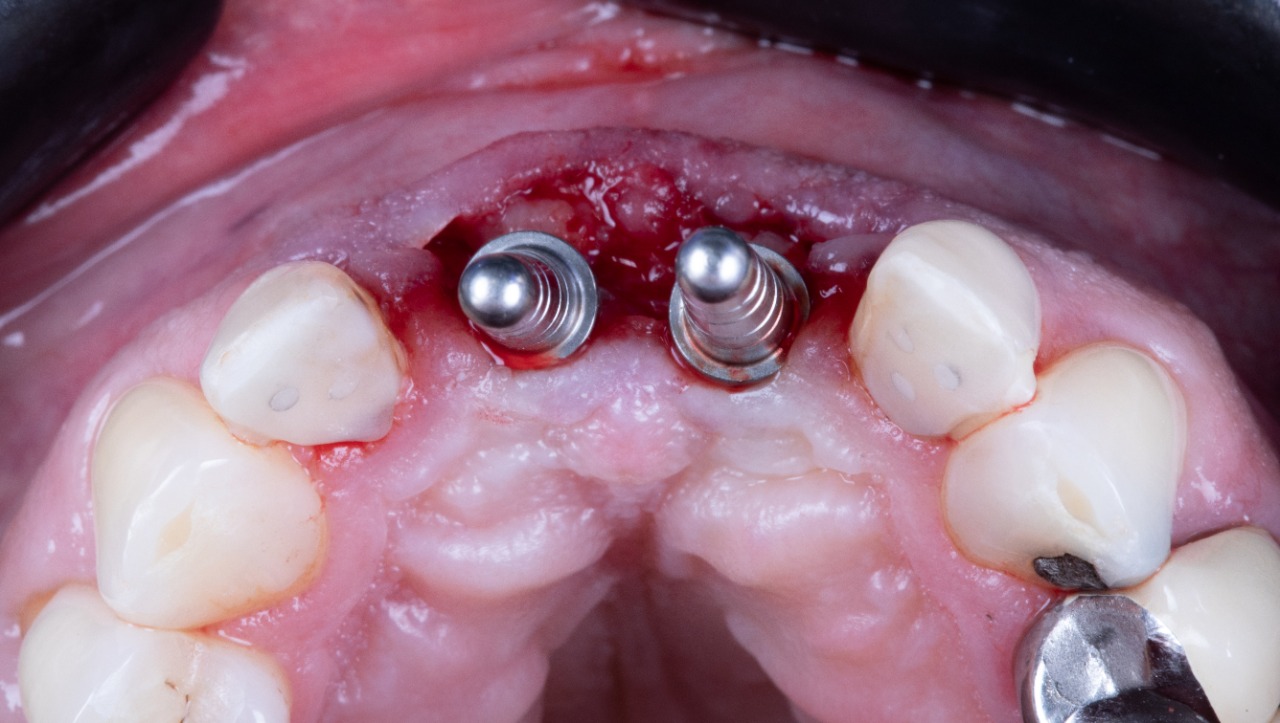
Prepared Osteotomies
Implant placement tool
Direction Indicators position verification (Front View)
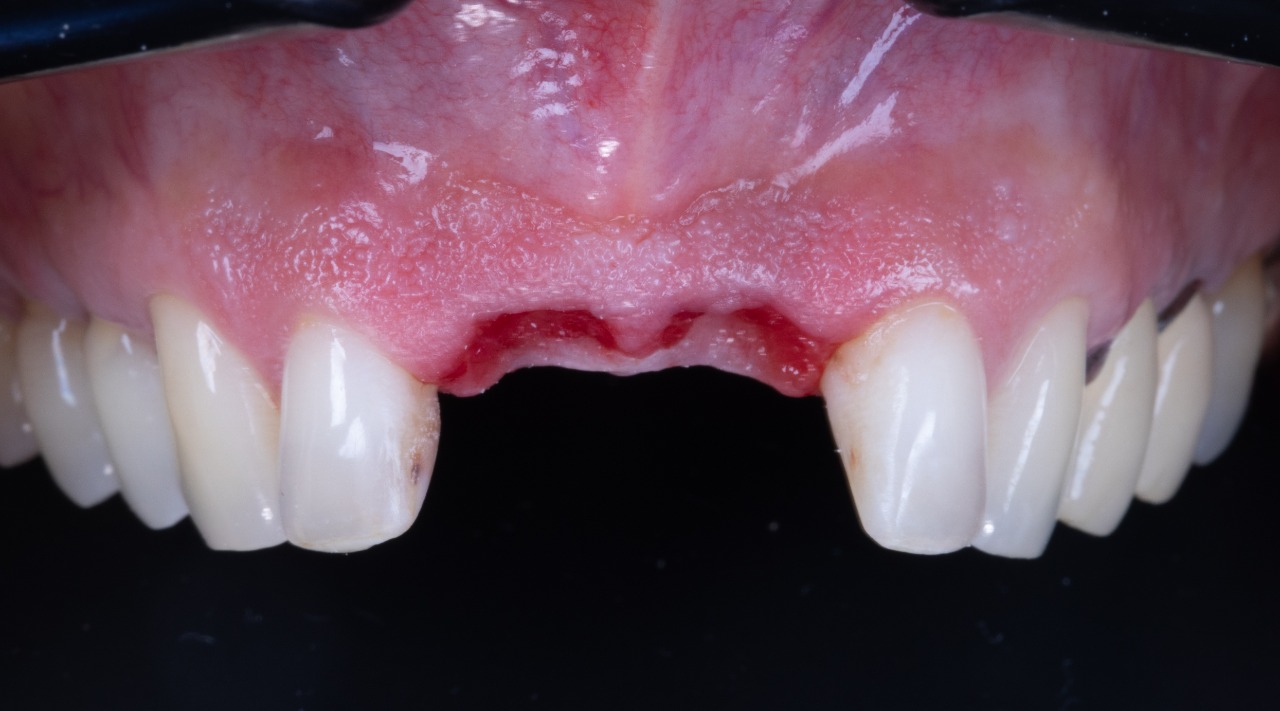
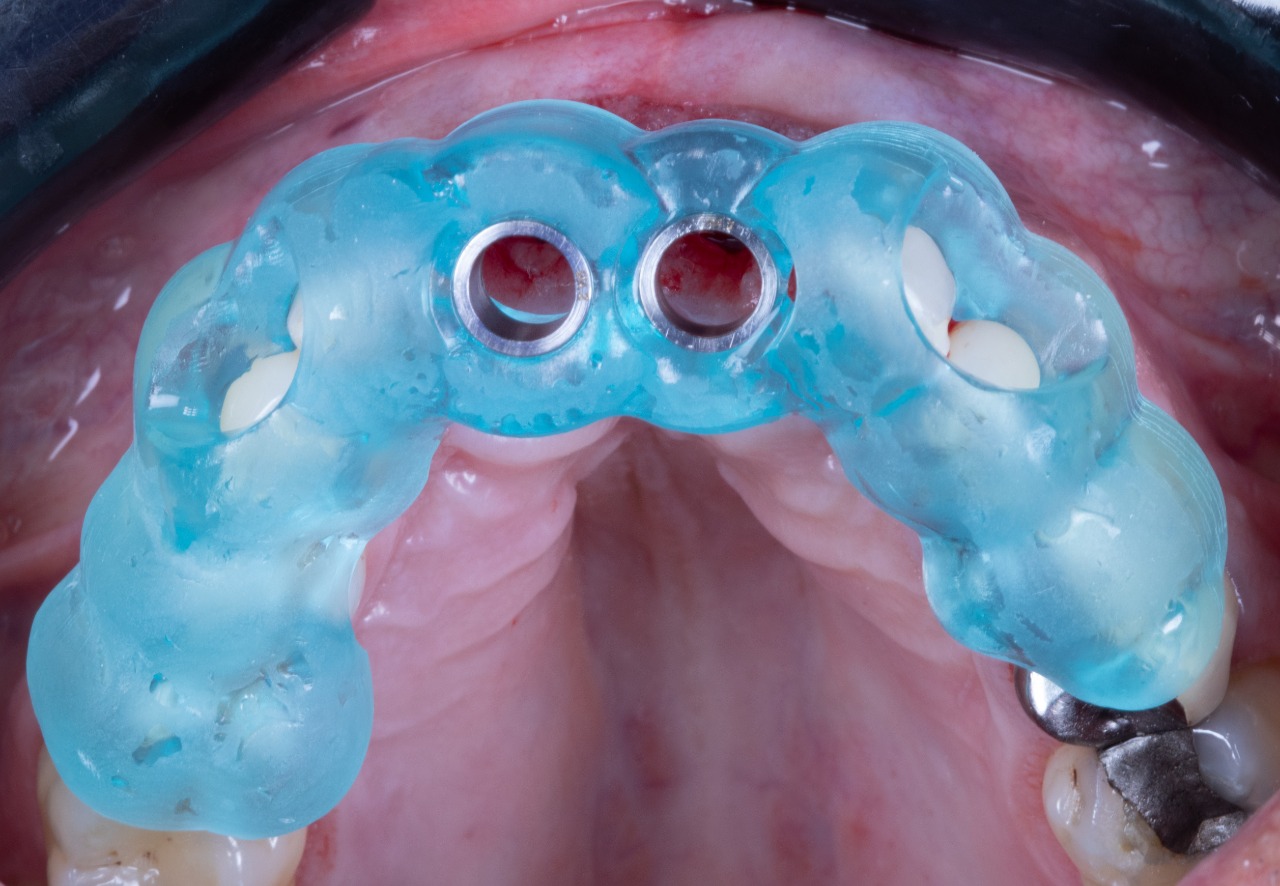
Implant Placement Guide Seated (Bottom View)
Implant Placement Tool, Cannon and Offset Sleeve assembly in the Implant Placement Guide
SIREAL Cannon Assembly with Implant Engaged and ready for placement
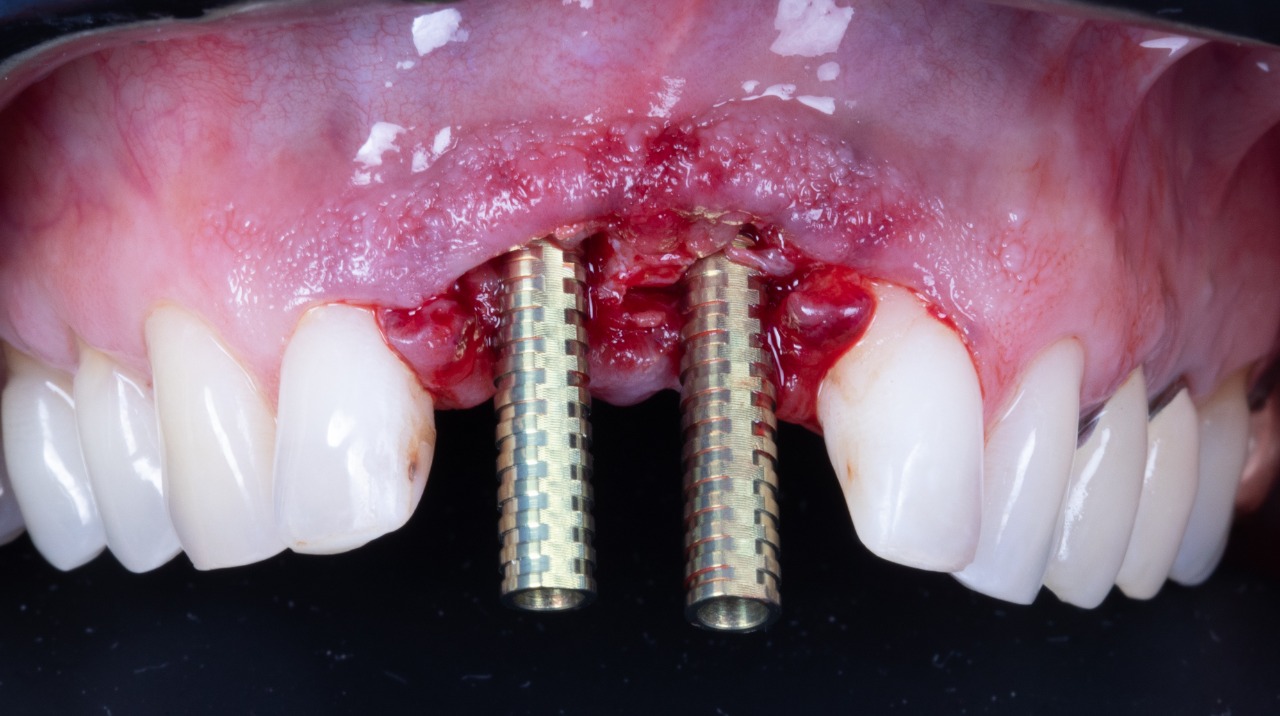
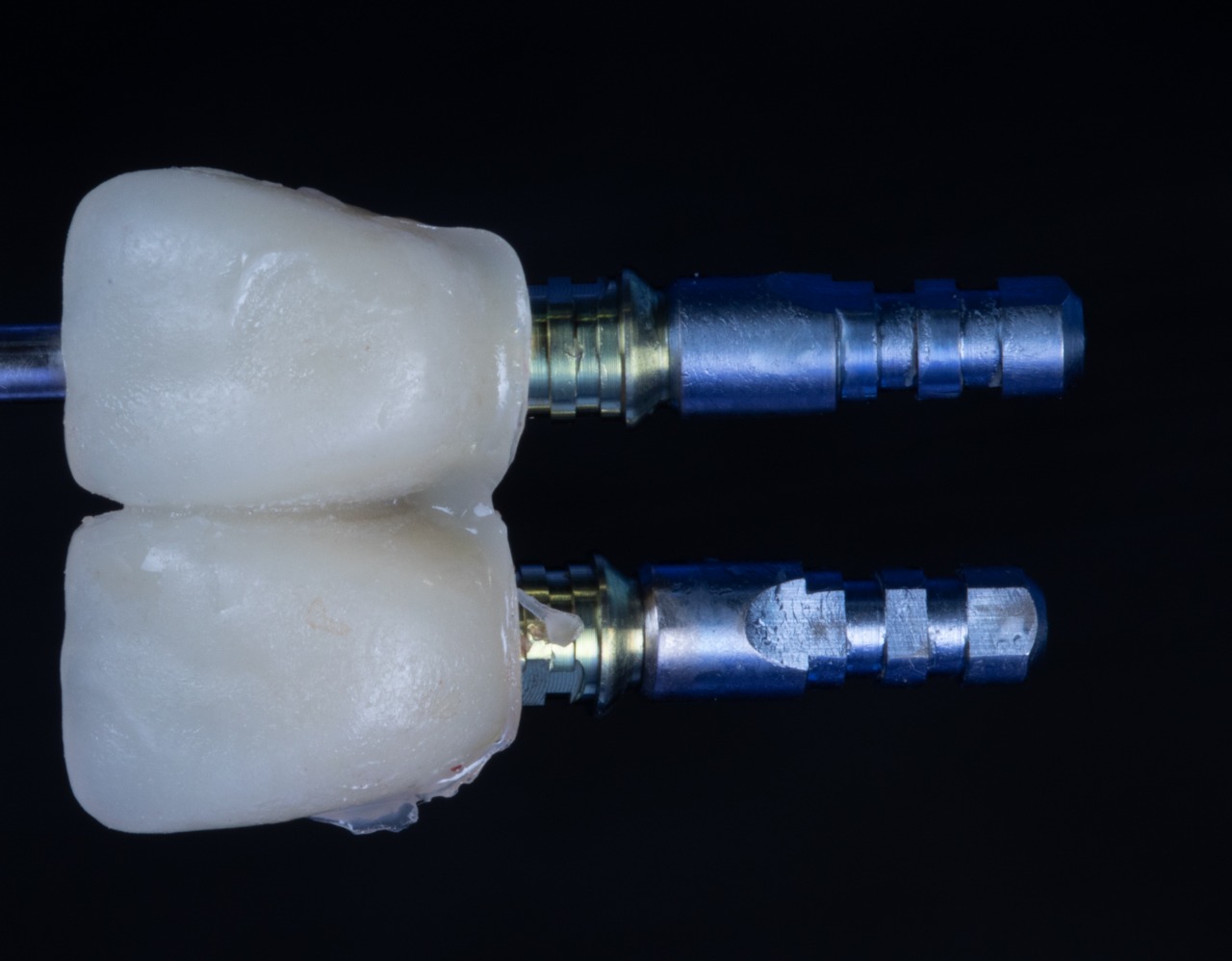
Temporary Titanium Cylinders (Bottom View)
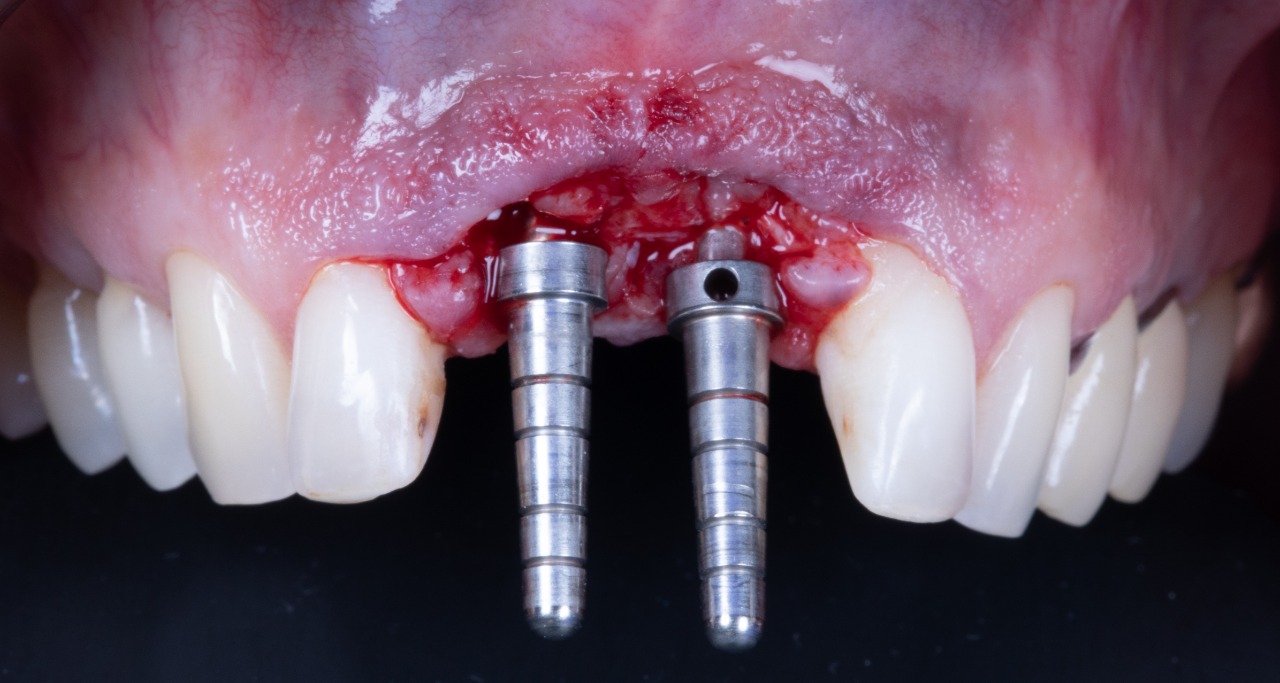
Deep Conical Implants placed at planned angulation and depth
Temporary Titanium Cylinders (Front View)
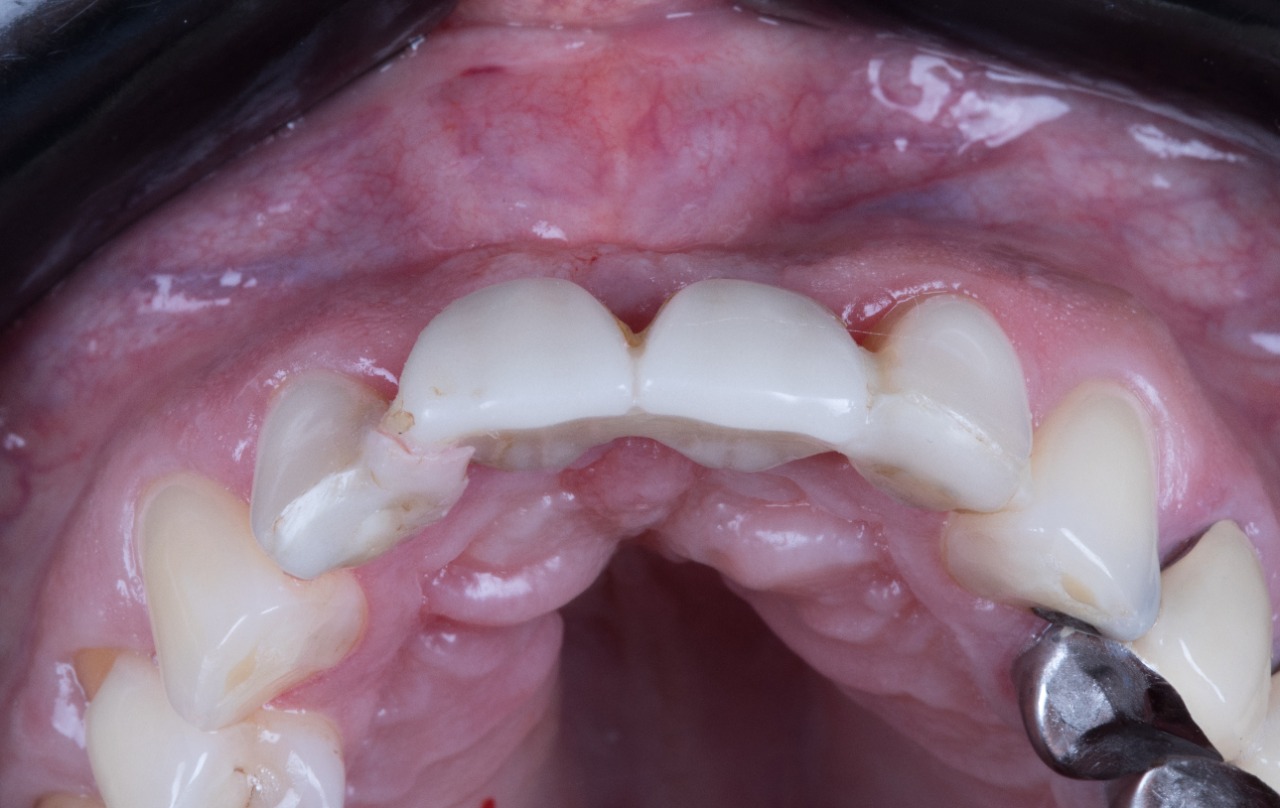
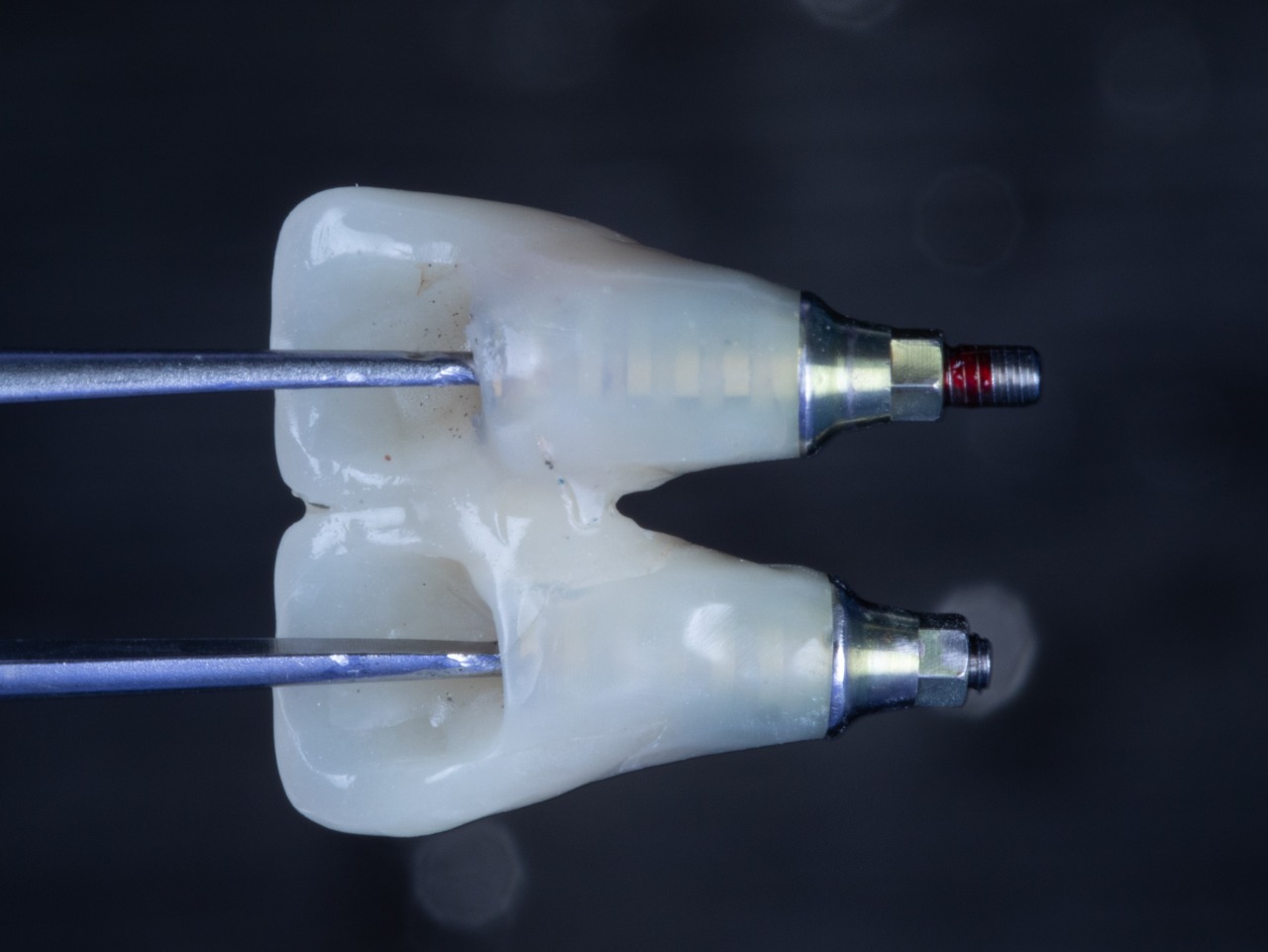
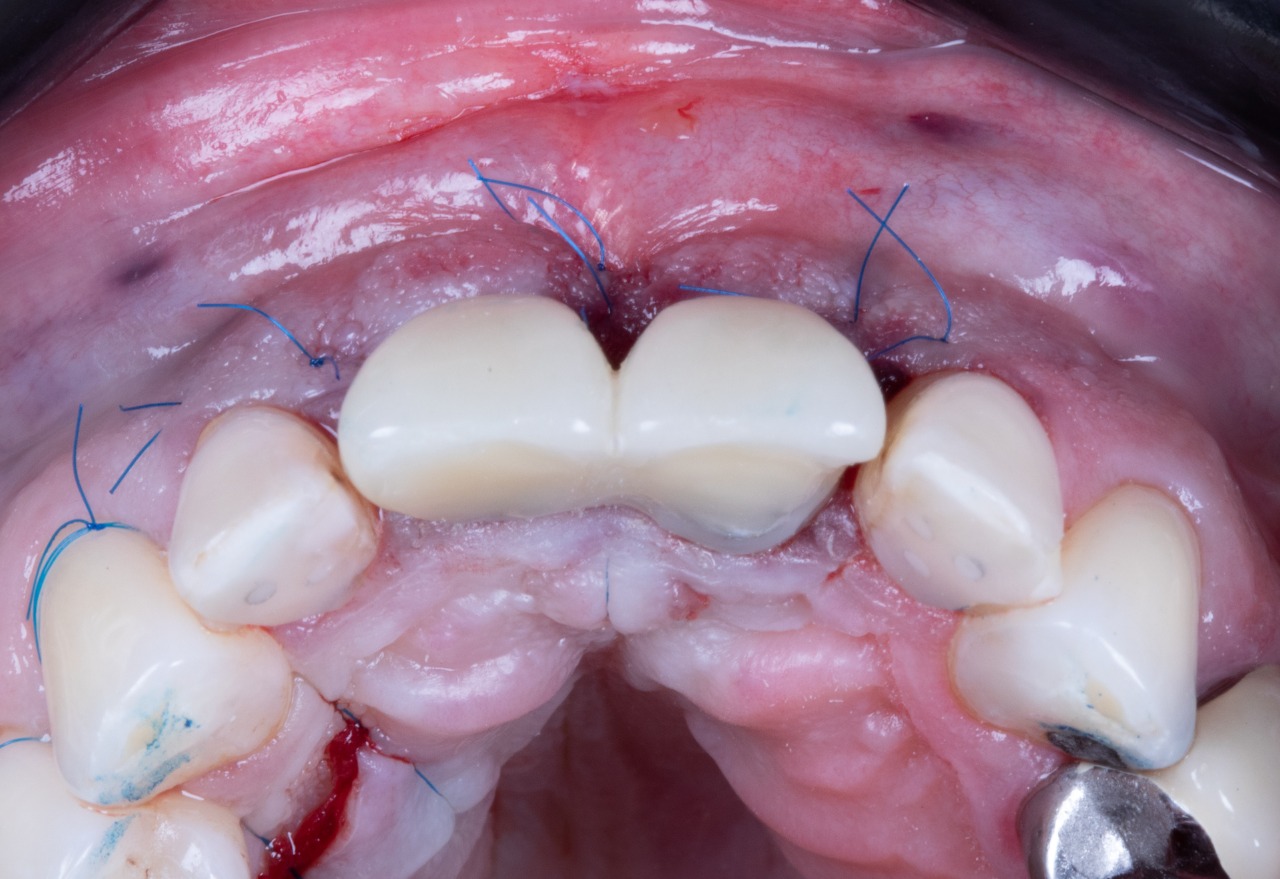
Temporary Crowns
Temporary Crown Placement (Bottom View)
Temporary Crown Placement (Front View)
Doctor-to-Doctor World’s Top One Hundred Recognition
Dr Stuart Lutton (New Life Teeth Edinburgh and Belfast) has achieved the Doctor-to-Doctor World’s Top 100 recognition for exemplifying leadership, excellence, and entrepreneurship in advancement of the healthcare industry.
He is humbled to be recognised globally for his work in full arch implant rehabilitation, patient care, research and development. He has introduced Brånemark priniciples to his dentistry clinic and is proud to be the UK & Ireland Brånemark Osseointegration Center.
The honorary recognition is an achievement award for exceptional services rendered to the arts and sciences of the healthcare professions.
The Global Summits Institute is a pre-eminent peer-to-peer organization in recognizing leading doctors in dentistry, optometry, pharmacy, chiropractic, surgery, and other medical specialties who exemplify clinical excellence, innovation, research, organizational leadership, and entrepreneurship in serving humanity and advancing the global healthcare industry.
Each member had been nominated by their peers and selected by a committee of their colleagues. Renowned clinicians, experts, key opinion leaders, surgeons, researchers, executives, innovators, and specialists from all inhabited continents were represented. 100 doctors were ratified.

Practice Efficiencies
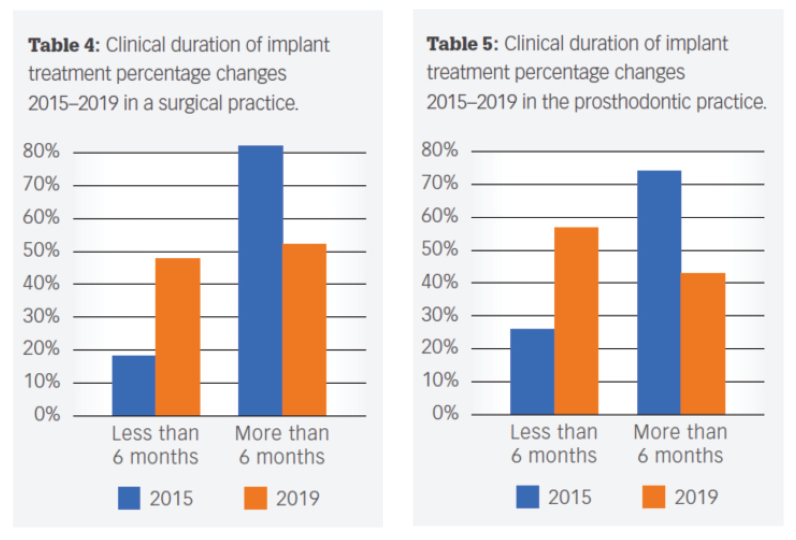
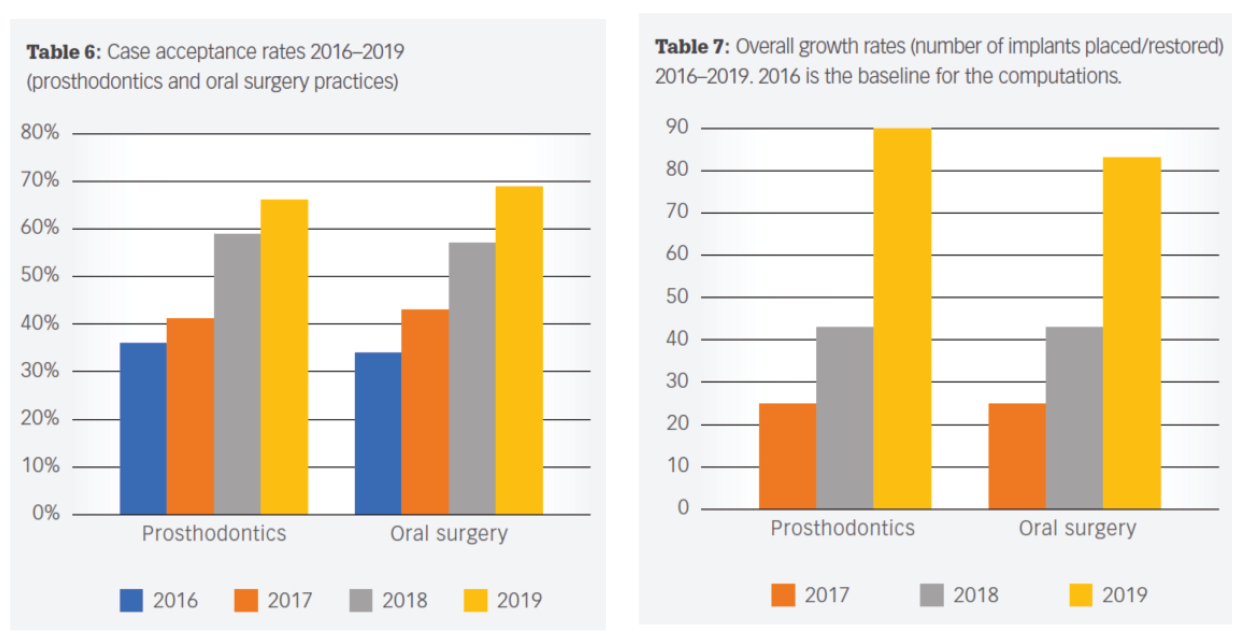
Today, predictable, accelerated implant treatment is more important than ever before. Recent research reveals that adapting the clinical practice with innovative treatment solutions provide significant benefits.
These benefits positively impact your patient’s satisfaction with treatment, as well as growing the number of implant procedures for surgical and restorative practices. The following innovations have led to real-world benefits for clinicians and their patients.
Patient-specific treatment
Site-specific implant designs allow for placing specific implants for specific clinical situations, as well as utilising the patient’s existing bone.
Southern’s site-specific implants include:
- Wider implants for immediate molar replacement.
- Narrower implants for thin bony ridges.
- Longer and shorter implants to support patient-specific implant planning.
- Angle correction at implant level.
- Inverted body shift designs for immediate anterior placement.
- Machined surface coronally for higher risk patients.
Improved practice effectiveness and efficiencies
Improved implant designs that achieve high insertion torque values allow for accelerated treatment by way of extraction and immediate implant placement.*
Increase in screw retained (versus cement retained) restorations
Using innovative implant restorative components increase the use of the screw retained restorations, resulting in decreased implant prosthetic complications and peri-implantation mucositis. **
Increased implant acceptance
By decreasing total treatment times without sacrificing implant success/survival patients are more likely to accept implant therapy.*
Increasing efficiency in Implant Dentistry
In a recent peer-reviewed article published in Dental Economics entitled Improved efficiencies in implant dentistry: Use of patient-specific accelerated therapy to drive practice growth, the authors, an oral surgeon and prosthodontist team, presented an analysis of implant cases throughout four years after incorporating site-specific implants into their treatment plans.
The results of this analysis were eye-opening. To quote from the conclusion: “Site-specific implants, together with appropriate protocols and team approach that the authors have adopted, have generated significant benefits.”
“More patients treated more definitely using faster protocols have allowed the authors to flourish even though the number of clinics offering less expensive dental implants continues to increase in areas adjacent to their practices.”
“The authors correlated the increased acceptance rates with several factors, including consistent, well-manufactured, precise implant and restorative components, multiple implant choices that fit a multitude of clinical situations; decreased treatment times; and a dedicated manufacturing/sales/administrative team.”
A full copy of the article can be downloaded here.
* Morris, G., Steinberg, M. and Drago, C., 2021. Improved efficiencies in implant dentistry: Use of patient-specific accelerated therapy to drive practice growth. DENTAL ECONOMICS.
** Staubli N, Walter C, Schmidt JC, Weiger R, Zitzmann NU. Excess cement and the risk of peri-implant disease – a systematic review. Clin Oral Implants Res. 2017;28(10):1278-1290.doi:10.1111/clr.12954
Research – What do we know about ceramic implants?
By Tamsin Cracknell and Kirosha Ramdan
Ceramic implants were first introduced by Professor Sami Sandhaus in 1967 to overcome the limitations associated with titanium implants, such as metal allergies and unpleasant aesthetic mucosal tissue discolouration in the aesthetic zone (1,2).
They were made of aluminium oxide, which had good osseointegration but insufficient biomechanical properties and fractured easily. Due to poor survival and success rates they were removed from the market in the early 1990s (1,2).
Zirconia dioxide ceramic implants were introduced by CeraRoot using yttria-stabilized tetragonal zirconia polycrystal (Y-TZP) in 2005. Systematic reviews indicate no significant difference between the biocompatibility of zirconia and commercially pure titanium dental implants (1–3).
Survival and success rates
Studies show that survival and success rates of zirconia implants are dependent on the implant design, surface characteristics, surgical protocols and prosthetic superstructures. Survival rates varied between 77.3% and 100% for 4 studies examining a monotype zirconia implant, where the follow-up period ranged from 2-5.9 years (1–3).
A 5-year analysis of one-piece zirconia implants with three different roughened surfaces loaded at four months showed an overall mean survival rate of 95%, specifically 92.77% for uncoated implants, 93.57% for implants coated with bioactive ceramic material and 97.60% for acid-etched implants (4).
In a more recent study assessing the Straumann PURE Ceramic implant, a 97.6% survival rate at 12-month follow-up was reported. Survival rate after 36 months was greater than 95% (7). According to the study, this is comparable to the survival rate of titanium implants placed with a similar protocol (3,4,8).
There are two case reports from the Z-Systems where the Z5C TL implant was placed in the molar area with a 1-year follow up. The report indicated good implant stability; healing was uneventful. Crestal bone level was maintained and gingival health was reported as excellent (5).
Strength and fracture
Zirconia implants exhibit favourable mechanical properties like low thermal conductivity, high flexural strength (900-1,200 MPa), fracture resistance, wear, and corrosion resistance (1,2). One study measured fracture strength of 277 N in the zirconia group and 165 N in the titanium group.
However, zirconia has low temperature degradation/ageing threshold. When in the presence of water or water vapour, a slow phase transformation occurs which, coupled with compressive stress, leads to an unwanted surface roughness that may result in progressive deterioration or microcracking (1,2).
The predominance of one-piece zirconia implants is a result of the material’s physical limitations; similarly, screw-retention and angulation of abutments presents a significant technical challenge. Geometric design of zirconia implants is important to avoid creating areas of stress concentration where cracks might propagate – this also precludes secondary reshaping using implantoplasty.
Failure in zirconia dental implants happens via rapid crack propagation directly through the implant head, compared to titanium implants where the prosthetic screw usually fails before the implant.
Zirconia is highly sensitive to its manufacturing processes. Different manufacturing approaches have been developed over the years to overcome these weaknesses, which has decreased fracture rates and increased survival. Several studies indicate that hot isostatic pressing or alumina enhancement of zirconia increases its favourable mechanical properties.
However, it is important to note that while survival rates of implants may appear high, biologic complications may occur as a result of forced cementation of restorations. Numerous manufacturers refer to their manufacturing process of ceramic implants as “innovative” and highlight the relationship between the breaking strength and the manufacturing process (4–6) – unfortunately, proprietary information on the precise manufacturing process is difficult to access.
Bone-to-implant contact
Zirconia implants with roughened surfaces produced by grit-blasting or acid-etching show a higher bone to implant contact but lower removal torque than their titanium counterparts (1,2,6). The bone to implant contact was significantly higher in four studies where zirconia and titanium implants were placed in rabbits. The zirconia implant had a micro-roughened surface created by selective infiltration etching compared to the titanium surface which was sandblasted and acid-etched (3).
Three studies in pigs showed significantly increased BIC values for acid-etched zirconia implants compared to electrochemically anodized titanium implants (3).
Peri-implant bone loss and soft tissue response
Peri-implant bone loss in a canine study was significantly higher in titanium implants than zirconia implants (1,2,7).
Studies comparing the bone levels of zirconia implants to titanium implants following the same surgical protocol found no significant difference in comparative follow-ups. For the zirconia implants the mean bone level 6 months after surgery was -0.88mm and -1.02mm 12 months after surgery. With titanium implants the bone level was -0.77mm +/- 0.93mm (6,8). At a 3-year follow-up of a study conducted by Straumann using the PURE ceramic monotype implant, bone loss was less than 1mm in 54.1% of sites. Bone loss was most pronounced in the first 6 months and remained relatively stable between 12-36 months, which resulted in an overall mean bone loss of 0.97mm at 36 months. Bone loss was more frequent in the maxilla than the mandible. When compared to the titanium Straumann Tissue Level Standard Plus at similar timelines, the difference was not statistically significant (6,8).
Peri-implant soft tissue response is similar to titanium and in some studies is reported to be superior. The bacterial adhesion on zirconia surfaces is comparable to titanium and in some studies lower on the zirconia surface than the titanium surface, suggesting the same level of tissue adhesion but with less plaque formation (2,7).
In a recent study with a 3-year follow-up, plaque was only reported on the adjacent teeth up until the 36th month where 3 of the 8 occurrences were reported on the implant side (3).
In vitro experiments provide evidence of no mutagenic and carcinogenic effects of zirconia implants (1,2).
Dental Implant Procedure
The ceramic implant is primarily used for restoration of a single tooth gap, with a natural tooth mesially and distally adjacent to the implant site (6,8). The implant requires a no-load healing process and in one study a thermostatic splint was used to protect the implant from micro-movement (6,8). Immediate loading of zirconia implants has a low survival rate: one study reported a 40% failure rate as opposed to a 3% failure rate when loading was delayed. It is important to note that insertion torque for zirconia implants is lower than titanium implants. It ranges from 20-30 Ncm for smaller-diameter implants to 45Ncm for a wider-diameter implant (3,8). This undoubtedly influences its susceptibility to failure under immediate loading.
The success rate of zirconia implants may be affected by augmented bone. A study reported a 97% implant success rate of one-piece zirconia implants in native bone, but 74% when placed in augmented bone (1,3). Any major bone augmentation, including bone transplantation, needs to be done at least 3 months prior to implant placement (4,6).
Conclusion
The designs, manufacturing methods, prosthetic attachments and surgical protocols of traditional titanium implants cannot simply be translated to zirconia implants. While they present significant benefits in terms of aesthetics and allergy prevention, their loading characteristics are quite different to titanium. More research into the effects of geometry, surface treatment, prosthetics and long-term survival is needed. A conservative approach should be taken when treating patients with zirconia implants in the absence of more conclusive results.
References
- Afrashtehfar K, Del Fabbro M. Clinical performance of zirconia implants: A meta-review. Journal of Prosthetic Dentistry. 2020 Mar 1;123:419–26.
- Cionca N, Hashim D, Mombelli A. Zirconia dental implants: where are we now, and where are we heading? Periodontol 2000. 2017 Feb;73(1):241–58.
- Roehling S, Schlegel Karl, Woelfler Henriette, Galhert Michael. Zirconia compared to titanium dental implants in preclinical studies-A systematic review and meta-analysis. Clinical Oral Implants Research. 2019 May;(5):365–95.
- Straumann® Ceramic Implant Systems – PURE and SNOW. Institut Straumann AG.
https://www.straumann.com/en/dental-professionals/products-and-solutions/dental-implants/ceramic-implants.html - Z-Publications – Z-SYSTEMS ceramic implants Switzerland. Z-Systems AG. https://zsystems.com/en/doctors/z-publications
- Gahlert M, Kniha H, Weingart D, Schild S, Gellrich N-C, Bormann K-H. A prospective clinical study to evaluate the performance of zirconium dioxide dental implants in single-tooth gaps. Clin Oral Implants Res. 2016 Dec;27(12):e176–84.
- Roehling S, Galhert M, Janner S, Meng B, Woelfler H, Cochran DL. Ligature-Induced Peri-implant Bone Loss Around Loaded Zirconia and Titanium Implants. https://pubmed.ncbi.nlm.nih.gov/30716147/
- Bormann K-H, Gellrich N-C, Kniha H, Schild S, Weingart D, Gahlert M. A prospective clinical study to evaluate the performance of zirconium dioxide dental implants in single-tooth edentulous area: 3-year follow-up. BMC Oral Health. 2018 Dec;18(1):181.
- About Z-Systems – Z-Systems ceramic implants Switzerland. Z-Systems AG.
https://zsystems.com/en/zsystems/about-zsystems - NobelPearlTM. Nobel Biocare AG. https://www.nobelbiocare.com/en-int/nobelpearl
- ZERAMEX® P6 implants – simple and efficient processes. ZERAMEX Dentalpoint AG. https://www.zeramex.com/en/dental_professionals/implant_systems_p6.php
- whiteSKY – The metal free implant. Bredent Group GmbH. https://www.bredent-implants.com/products-solutions/sky-implant-system/one-piece-implants/whitesky/
- Medical Instinct® – BoneTrust® Implant systems. Medical Instinct® Deutschland GmbH. https://www.medical-instinct.de/en/bonetrust-implant-systems/
- TAV Dental Zirconia Implants. TAV Dental. https://tavdental.com/
Southern Staff Achievement
One of the Southern Implants engineers, Alexandra Stuart-Smith, won 2nd prize at the Academy for Osseo-Integration’s virtual meeting for her presentation on Southern’s ASC (Angulated Screw Channel) system which she is developing.
Alex was born and raised in Cape Town, South Africa. She holds an MSc in Biomedical Engineering from Imperial College London and a BSc in Chemical Engineering from the University of Cape Town. She is currently a Design Engineer at Southern Implants and pursuing the career she envisaged while completing her Masters.
The following is the abstract from her award-winning article:
Title:
Angulated Screw Channel Solution for Increased Preload
Authors:
Stuart-Smith, Alexandra

Abstract:
Angulated Screw Channel Solutions have gained a reputation of being associated with “screw loosening” resulting from a decreased preload in the retaining screw due to the angulation. Further complications include interface distortion and reduced torque transfer. This study aimed to propose a new Angulated Screw Channel Solution, utilising high strength materials and optimum dimensions, which can take up to 45 Ncm torque and hence generate significantly higher preload than current solutions and preload equivalent to or greater than a non-angulated screw channel at the recommended torque (32-40 Ncm).
In order to test the newly designed ASC solution, preload testing was performed on titanium M2 screws with the optimally designed driver. Torques ranging from 30-45 Ncm were applied to the ASC solution at angulations from 0° to 25°.
Only a maximum of 3% reduction in preload was observed at the 10° condition with many of the results exceeding or equaling that of a non-angulated solution. For the 20° and 25° conditions, a preload drop of maximum 10% was observed. These trends are consistent with the theoretical torque drop-off expected as the angulation increases. At all angulations, a tightening torque of 45Ncm was achieved and no interface damage or distortion was observed on the screw or the driver. The preload values achieved at 45Ncm for all angulations, 10°, 20° and 25°, were equal to and up to 1.3 times greater than the preloads achieved at torques within the recommended torque range for the 0° condition.
By utilising high strength materials and optimising the dimensions, a new Angulated Screw Channel Solution was developed that can achieve preloads equal to and up to 1.3 times greater at a torque of 45 Ncm for all angulations than the preloads achieved within the recommended torque range (32 – 40 Ncm) for the 0° condition.


UPCOMING EVENTS
United Kingdom:
London Dentistry Show at Olympia
17-18 September 2021
ADI Team Congress – Manchester
26-28 May 2022
USA:
American Academy of Periodontology (AAP)
November 4-7, 2021
Miami Beach, FL
More Information
American Academy of Maxillofacial Surgeons (AAOMS) Dental Implant Conference (DIC)
December 2-4, 2021
Chicago, IL
More Information
Greater New York Academy of Prosthodontists
December 3-4, 2021
New York City, NY
More Information
France:
Implantology Academia Aix en provence
September 13-16, 2021
More Information
56e Congrès de la SFSCMFCO – Besançon
29 September – 2 October
More Information
Congrés du CLIP Pont à Mousson
September 30, 2021
More Information
Syfac PRF Paris
September 30 – 1 October, 2021
More Information
ADF Paris
November 23 – 27, 2021
More Information
Syfac PRF Nice
December 9 – 10, 2021
More Information
ISO AGEN
October 9, 2021 and December 11, 2021
More Information
France
Sitour Marseille
October 6, 2021
Zygo Meeting Paris
October 14, 2021
More Information
Dr Renouard’s Training Vauvert
October 15-16, 2021
More Information
North America
SAVE THE DATE!
Southern Implants North America Conference
December 9-12, 2021
Delray Beach, FL
South Africa
SAVE THE DATE!
Southern Implants International Forum 2022
March 20-23, 2022
South Africa