Table of Contents
Graham Blackbeard,
Founder and Managing Director
Message from Graham
Welcome to the first issue of “Southern Implants In Focus” for 2020
“The journey is just as important as the destination.” I read this statement in a recent letter and reveled in the truth of it. The Southern Culture is one rightfully fixated on the destination; ensuring our company and products are meeting our responsibilities to our customers, their patients, to society and also our employees. We push hard, forgetting at times to enjoy the journey.
Good may be good enough for some, but we want more than that. To find that balance between our responsibility and being better than good, but still enjoy the Journey has been our challenge and one we will address in the year to come.
We began 2019 by restructuring our Marketing Team and aligning our skillsets to enable the team to create high-quality content in a timely manner. I can confidently say we’ve seen numerous examples across our markets of these efforts bearing fruit. We continue to promote our company story and we look forward to continuing the momentum into 2020.
2019 also proved to be a stand-out year for new product introductions with the successful US launch of the INVERTA® Implant in June at the ISPRD Meeting in Boston. This launch included beta test groups from around the world documenting cases for analyzation. An increasingly large part of dental implant regulatory activity is devoted to gathering product feedback from real users in typical clinical settings. Formal clinical trials are excellent for assessing specific performance aspects, but seldom reflect real-world conditions or the diverse patient characteristics seen in practice.
Increasing our focus on Digital Dentistry was another phenomenal milestone in 2019. SIDigital Solutions brought forth prosthetic libraries released for 3Shape, Exocad and Dental Wings, allowing the digital team to create a seamless end-to-end experience for Southern Customers. There were certainly challenges due to the diversity found in the combination of software and hardware used by different customers. But the team overcame it with two successful library updates, which were the product of joint efforts between the Southern Digital Team and valued Southern Customers. These updates included changes to tolerances and base file designs, as well as orientation adjustments for Digital Lab Analogues, improvements with the Passive Abutment and the introduction of the Passive for the Piccolo Implant. The development of the SIGuided Kit was also a milestone. All of these developments and updates have been beneficial to the digital workflow and we are excited to introduce these to you. Read more about Southern’s digital offering from a clinician’s point of view in this article by Professor P.O. Östman
I hope you enjoy this issue of Southern Implants InFocus Newsletter. As always, thank you for your support and collaboration in helping us provide innovative solutions. Wishing you all the best for this new decade.
SIGuided – Southern Implants guided surgical solution
Pär-Olov Östman, DDS, PhD, MD
Falun, Sweden
For some time, guided solutions have grown in popularity, and many clinicians desire to have the option to approach treatment planning using planning software and guides. Patients are also becoming increasingly discerning, requiring outstanding aesthetic results with a minimum number of appointments at the practice. To meet these expectations, precision and seamless collaboration within the treatment team and network is crucial. An integrated implant planning workflow offers innovative benefits for implant planning and prosthetic rehabilitation, combining this with an interdisciplinary team approach can increase treatment efficiency and save chair time during the surgical procedure.
The digital workflow has an increasingly important role to play in dentistry today. SIDIGITAL, Southern Implants’ digital solution for CAD/CAM procedures has been successfully used for a number of years. SIGUIDED is the most recent development in Southern Implants digital workflow, allowing for fully guided implant placement of both External Hex and PROVATA® Implants.
Why is there a need for guided surgery? Placing an implant from a peri-apical radiograph alone can, at times, present challenges. Studies have shown that in approximately 20% of cases where implants were placed in the anterior, an apical perforation can be seen. Moreover, it can, in many cases, be difficult to ensure that there is a 1.5mm space mesial/distal between the implant and the adjacent tooth, as well as 2mm space between the implant and the buccal/palatal bone without the assistance of 3D planning tools and guides.
An overview of guided surgery:
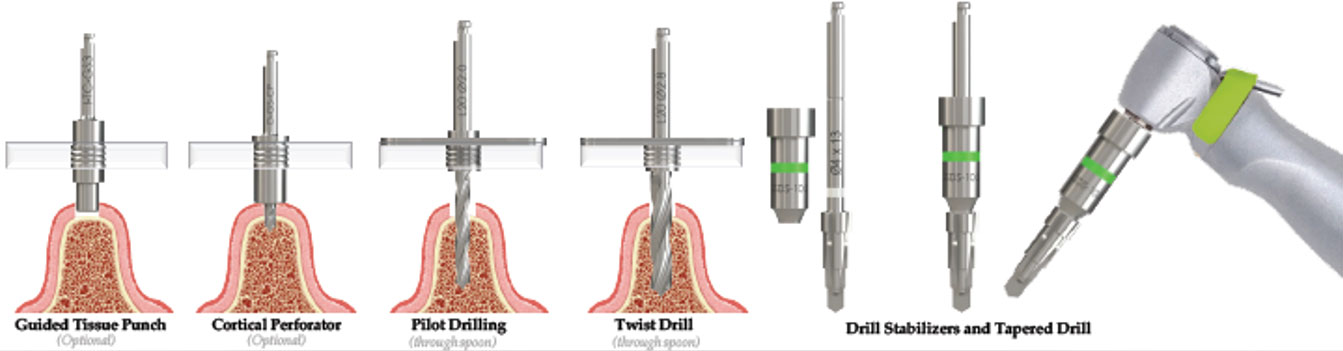
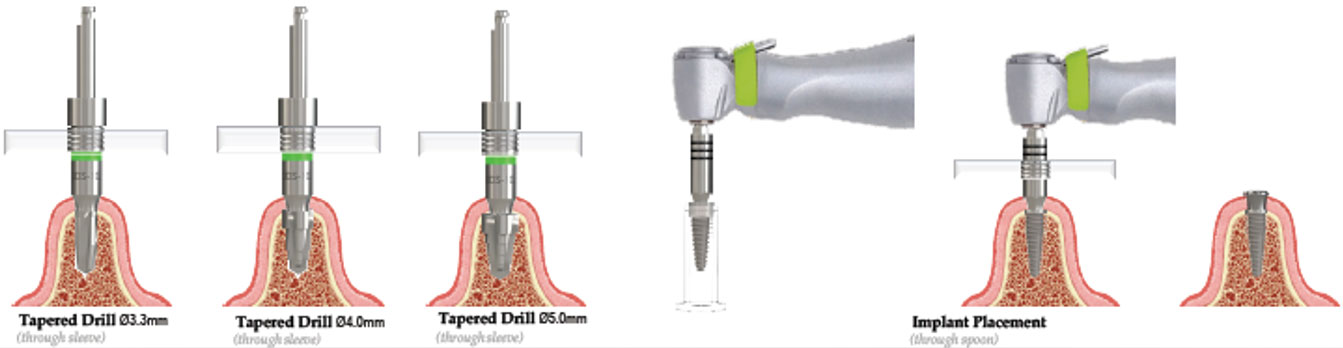
CBCT scanning of the patient, is a 3D investigative tool to determine bone height, bone thickness, and important anatomical landmarks to avoid. The intraoral scan is used to create a digital representation of the oral environment. The CBCT and intraoral scan are imported into planning and guided design software, allowing the practitioner to plan implant placement in a 3D environment by means of the CBCT scan. The intraoral scan gives the added advantage of an exact digital representation of the patients oral environment to design a surgical guide. A virtual abutment/prosthetic screw axis hole is placed in the software to determine if the planned implant position will provide the best prosthetic outcome. This pre-planning oftentimes results in superior prosthetics and well-controlled work by the dental technician.
In this context, digital implant planning and guided implant surgery based on three-dimensional radiographic data and the digitised intraoral surfaces can be of excellent service. These tools provide valuable information and permit stringent backward planning to optimise implant placement and prosthetic results, improving the safety and efficiency of the surgical procedure and rendering the restorative outcome more predictable in terms of function, biology and aesthetics.
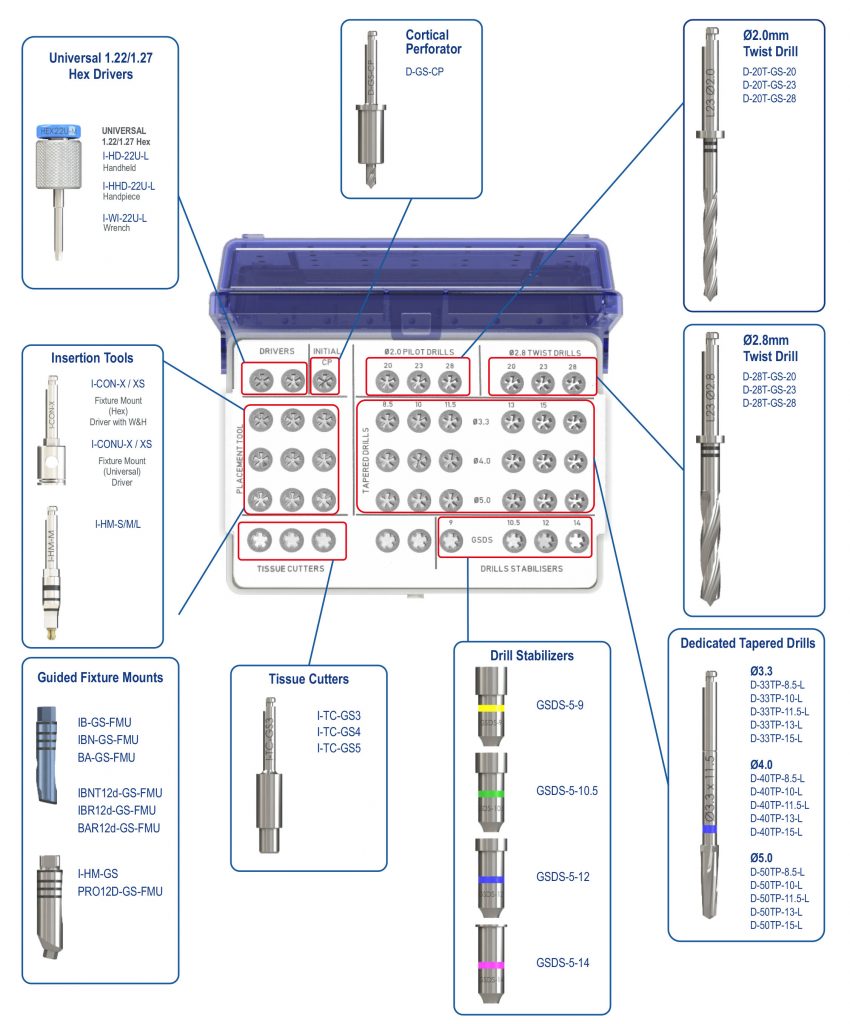
Southern Implants Guided Solutions were developed to be a flexible treatment solution enabling fully guided, pilot guided, bone and soft tissue supported, or tooth supported for full adaptability. The SIGuided Surgical Tray and components were designed to deliver high precision with as few components as possible (SIGuided Kit Schematic below). The kit includes four drill-stabilizers, offering different prolongations with high precision in vertical, as well as horizontal positioning, yet keeping the componentry to a minimum.

The drill stabilizer is placed over the tapered drill shaft and secured with the hand piece. The stabilizer is used to guide the drill through the guide sleeve. The drill stabilizers have set stopper heights which are dependent on the guide offset. Drill stabilizers are colour coded corresponding to prolongation and offsets for ease of identification.
Also included in in the SIGuided Surgical Kit are tissue cutters, dedicated tapered drills, ø2mm, ø2.8mm drills, cortical perforator, long and short spoons, fixture mounts and placement tools if a fully guided protocol is desired. The dedicated long drill included can eliminate the need for drill extensions. The kit may also be used to place the same implants conventionally reducing the need for multiple kits.
 Clinical Research – The core of product development
Clinical Research – The core of product development
Nazalus Implants
Extra-Long Nasal Wall-Directed Dental Implants For Maxillary Complete Arch Immediate Function: A Pilot Study. Oral Maxillofacial
Nicoli G, Ferraris P, Jensen OT.
Surg Clin N Am 31 (2019) 349–356.
First clinical results of fixed full-arch restorations on 4-6 implants using both straight and 24° implants affixed in the nasal floor and wall
In this first clinical publication reporting on 33 patients receiving 53 Nazalus implants as part of 4-6-implant maxillary full-arch restorations, co-creators Nicoli, Ferraris et. al. reported 98% success at 16-61 months follow-up. A total of 27.3% implants had to be loaded with a delayed protocol due to primary stability <30Ncm. Sinus floor grafts were performed for 58.3% of implants, according to the recommended protocol of grafting when crestal bone height is <3mm. No other complications occurred. The authors’ conclusion was that Nazalus Implants are a less-invasive, predictable alternative to zygomatic or pterygoid implant treatments.
Short Implants
Posterior atrophic jaws rehabilitated with prostheses supported by 6-mm-long 4-mm-wide implants or by longer implants in augmented bone. Five-year post-loading results from a within-person randomised controlled trial.
Felice P, Pistilli R, Barausse C, Piattelli M, Buti J, Esposito M.
Int J Oral Implantol (New Malden). 2019;12(1):57–72.
Felice et. al.
Placement in atrophic mandible/maxilla compared to 10mm implants with concomitant bone grafting.
From the University of Bologna published five-year results in a split-mouth randomised controlled trial comparing short implants (length 6mm) with standard-length (10mm) implants placed in augmented bone in the posterior atrophic jaw. Forty patients (20 mandibles, 20 maxillae) received between one and three short implants on one side of the jaw and one to three standard implants contralaterally. The implants were definitively loaded after 4 months. Unfortunately, eight patients dropped out of the study before the five-year recall, but of the remaining patients, there were no statistically significant differences in implant failures. Nineteen complications occurred at augmented sites versus five complications at short-implant sites. In the mandible, patients with 6mm implants lost an average of 1.34±0.35mm bone, versus 2.11±0.59mm for 10mm implants with grafting. In the maxilla, 6mm implants lost an average of 1.52±0.47mm versus 1.85±0.51mm for 10mm implants. Both differences were statistically significant in favour of short implants.
Co-Axis Implants
Immediately restored single implants in the aesthetic zone of the maxilla using a novel design: 5-year results from a prospective single-arm clinical trial.
Ma S, Tawse-Smith A, Brown SDK, Duncan W.
Clin Implant Dent Relat Res. 2019 Apr;21(2):344–51..
CO-AXIS: Immediate loading in the aesthetic zone with full ceramic crowns.
A study from Otago presents five-year clinical results from a prospective trial of 27 patients receiving single Co-Axis 12° implants in the anterior maxilla. Implants were temporised within four hours of placement and restored with definitive screw-retained zirconia abutments at eight weeks post-placement. Sixteen participants attended five-year recall, and results showed that mean marginal bone loss between one and five years was 0.1±0.25mm overall. The mean ISQ at five years was 69.9. No prosthodontic maintenance issues had occurred between one= and five-year follow-up.
INVERTA Implants
INVERTA Registry
An increasingly large part of dental implant regulatory activity is devoted to gathering product feedback from real users in typical clinical settings. Formal clinical trials are excellent for assessing specific performance aspects, but seldom reflect real-world conditions or the diverse patient characteristics seen in practice.
“Post-Market Surveillance” (PMS) is required by most international regulatory authorities and involves amassing and analysing huge amounts of data relating to patient demographics, surgical and prosthetic protocols, and narrative feedback from doctors and laboratories.
 Southern Implants, together with South African software company Ubiquitech, has developed a bespoke Online Registry for use by clinicians belonging to the Beta Testing Group of the new INVERTA Implant. This group of some 40 periodontists, prosthodontists, maxillofacial surgeons and oral surgeons was hand-selected from the USA, UK, Australia and South Africa based on their experience with Southern Implants products and scientific research. Participation includes completing at least five INVERTA Cases, reporting a wide range of parameters via the Online Registry.
Southern Implants, together with South African software company Ubiquitech, has developed a bespoke Online Registry for use by clinicians belonging to the Beta Testing Group of the new INVERTA Implant. This group of some 40 periodontists, prosthodontists, maxillofacial surgeons and oral surgeons was hand-selected from the USA, UK, Australia and South Africa based on their experience with Southern Implants products and scientific research. Participation includes completing at least five INVERTA Cases, reporting a wide range of parameters via the Online Registry.
The Registry aims to replace traditional paper-based Clinical Research Forms (CRFs), allowing both clinicians and administrators to store, view and extract data in digital format for use in publications and design development. Similar platforms are commercially available but tend to be designed for large pharmaceutical trials; hence, the decision was made to build a brand-new platform from the ground up that is appropriate for medical device trials, and specifically implant dentistry.
Access to the Registry is secure and password-protected, and participants can only view and edit cases performed in their own practices. The format consists of sequential digital forms covering each step of the treatment programme. Images such as CBCTs and clinical photographs can be uploaded for comparison between timepoints, and built-in logic checks mitigate the need for pre-analysis data cleaning. Registry administrators can extract data directly into any statistical analysis software.
Says Ubiquitech Director Rob Pitman: “The system complies with industry best practices and standards for information security, scalability and reliability. It is accessible via any modern web browser (such as Internet Explorer, Google Chrome or Mozilla Firefox). Users need only a reliable Internet connection to upload and record their data.”
In this way, large amounts of data from all around the world can be stored and accessed securely and indefinitely. Digital algorithms negate data pre-processing and filtering, which speeds up analysis. Physical storage needs are minimised, and the platform is scalable to any number of participants and projects. Southern Implants plans to roll out this platform for all of its products, leading PMS into the digital age.
UK INVERTA Beta Registry Launched
After launching in the United States of America in June 2019, the UK’s beta group was launched in August. Below are scenes from the first of its kind Beta Registry.
Drs. Hennie van Jaarsveld and David Offord during a hands-on session.
Dr. Duncan Robertson during a hands-on session.
Dr. Stephen Chu, co-designer of the INVERTA Implant, dialing in from USA to share his insights into this novel implant. “It’s all about the biology”.
Maxillofacial Conference in Cape Town
The 2019 SASMFOS conference was held in Cape Town with two days of workshops preceding the academic programme. Cadaver workshops were held at Tygerberg Hospital and delegates had the opportunity to perform flaps and other surgical procedures, including placing Zygomatic/Zygan Implants.
Speakers included Prof. P.O. Östman (Digital Workflow, Immediate Placement Molar Socket, Anterior Socket using a Body-Shift™ Implant), Drs. Michael Michael and Noland Naidoo (Prosthetic Reconstruction), Mr. Martin Danford (Perforated Flap Design) and Dr. Greg Boyes-Varley (lectures and a hands-on session with models using Nazalus, MAX, INVERTA, Zygomatic, Oncology and Zygan Implants).
American Academy of Esthetic Dentistry (AAED)
The theme of this annual meeting was Disruptive Dentistry and the Opportunities Ahead. With its long history and Innovative Treatment Solutions, there isn’t a better manufacturer to be a part of this unique event than Southern Implants! The INVERTA Co-Axis® Implant created a lot of buzz from this esteemed group of aesthetic driven clinicians who may have heard about this new implant from the launch earlier in the year at ISPRD in Boston. There was a “Wow” Roster of main podium speakers and moderators, some of which included Drs. Stephen Chu, Dennis Tarnow, Costa Nicolopoulos, Petros Yuvanoglu, Howard Gluckman and Barry Levin.
Pikos Symposium
Southern Implants stood out at the Pikos Symposium October 10-12. Drs. Costa Nicolopoulos and Petros Yuvanoglu educated attendees during our All on X Hands-on Workshop utilizing existing bone with Co-Axis and MAX Implants. The next day the dynamic duo took the main stage to share that message again to all symposium attendees, driving a lot of traffic to the Southern Team. Dr. Nicolopoulos took the main stage again teaching what to do for a failed full-arch case. Last, but certainly not least, Dr. Carlos Aparicio presented the ZAGA approach and when questioned about what Zygomatic System he used, he declared Southern Implants Zygomatic Implants!
American Academy of Maxillofacial Prosthodontists (AAMP)
Dr. Claudio Brenner provided an insightful look into the treatment of Oncology Patients. This was the first year Southern Implants North America carried the Steco Product Line of Titanmagnetics® at this meeting to complement our Extra-oral Implants.
American Academy of Periodontology (AAP)
The trailblazing INVERTA story continued at AAP. Dr. Robert del Castillo presented to an excited crowd during Southern’s corporate forum. Dr. Tarnow lectured from the main podium, and had a never-ending line at the Quintessence book signing of his new book “The Single Tooth Implant”. Be sure to check out the end of chapter 2 to see Co-Axis and INVERTA.
First UK and Ireland Brånemark Osseointegration Centres – New Life Teeth
 Congratulations to Dr. Stuart Lutton, principal of New Life Teeth in Belfast and Edinburgh, and his team. These two practices have been awarded the most prestigious accolade in implant dentistry – they are the first UK and Ireland Associated Brånemark Osseointegration Centres (ABOC).
Congratulations to Dr. Stuart Lutton, principal of New Life Teeth in Belfast and Edinburgh, and his team. These two practices have been awarded the most prestigious accolade in implant dentistry – they are the first UK and Ireland Associated Brånemark Osseointegration Centres (ABOC).
New Life Teeth is one of the worldwide clinics selected based on clinical excellence in dental implantology with an emphasis on their patient care and sound biological and clinical principles to carry this accreditation. Southern is proud to be associated with these amazing practices.

Professor Dale Howes
 Prof. Dale Howes, founder member of the P-I Brånemark Institute in South Africa, has accepted a position as Associate Professor of Prosthodontics at the School of Dentistry, Faculty of Medicine and Health, University of Sydney.
Prof. Dale Howes, founder member of the P-I Brånemark Institute in South Africa, has accepted a position as Associate Professor of Prosthodontics at the School of Dentistry, Faculty of Medicine and Health, University of Sydney.
He is the current Vice President of the ISMR (International Society for Maxillofacial Rehabilitation), board member of the ICP (International College of Prosthodontists), invited fellow of the International academy for Oral and Facial Rehabilitation (IAOFR) and serves on the North American working group for Advanced Digital Technology in Craniofacial Reconstruction. He has received fellowships from the College of Dentistry of South Africa and the International College of Dentists.
Prof. Howes is a past president of the Academy of Prosthodontics of South Africa. In addition to being founder member of the P-I Brånemark Institute of South Africa, he is also a member of the Face Value Foundation Trust.
Introducing Southern Implants Ibérica
 On 1 October 2019, Southern Implants Iberia, a new Subsidiary, opened its doors. Leading the Subsidiary is Mr. Jordi Demestre, who has been working in the implantology market for the past 19 years before joining Southern Implants in October 2018.
On 1 October 2019, Southern Implants Iberia, a new Subsidiary, opened its doors. Leading the Subsidiary is Mr. Jordi Demestre, who has been working in the implantology market for the past 19 years before joining Southern Implants in October 2018.
Based in Barcelona, Spain the Southern Implants Ibérica office will start with four team members. Jordi says that “our aim will be to listen to customers and their needs and will focus on providing a five-star service. We aim to sell solutions, not implants, focusing on advanced treatment solutions like full-arch and zygomatic cases and solutions for the aesthetic zone.”
Southern Implants Ibérica first major event was at the International Sepes congress in Barcelona in early October. Be on the lookout for more exciting news coming from this new Subsidiary.
South Africa
Advanced Implant Dentistry Course
February 7-9, 2020
P-I Brånemark Institute South Africa
Irene, South Africa
Southern Implants International Forum – Balancing Facts with Elegant Thinking
March 29 – April 1, 2020
Fairmont Zimbali Conference Centre
KwaZulu Natal, South Africa
Spain and Portugal
Guided Bone Regeneration Symposium 2020
Bone Biomaterials & Beyond Academy
Sevilla, Spain
March 28, 2020
UK & Ireland
UK and Ireland Forum 2020
June 4-6, 2020
Mere Golf and Spa Resort Knutsford
United States of America
Southern California Consensus Symposium (SCCS)
February 6-8, 2020
JW Marriott Los Angeles
Academy of Osseointegration (AO) 35th Annual Meeting
March 19-21, 2020
Seattle, Washington