
The
MAX
implant
designed to provide a treatment solution for molar tooth replacement

Prof Dr Pär-Olov Östman & Dr André Hattingh
A common need for dental implant treatment is replacement of a failing molar tooth.
The size and shape of the multi-rooted socket is often not suited to the placement of a typical implant, resulting in compromised implant positioning or poor primary stability. This may result in a waiting period of 3-4 months to allow for healing before attempting to place an implant. The healed site will often present with reduced bone height, resulting in the need for augmentation, especially in the maxilla. This leads to further lengthening of treatment time with increased cost and complexity.
Southern Implants’ MAX implant is designed to provide a treatment solution to overcome the challenges of placing an implant in a multi-rooted molar socket in mandible or maxilla.

It is intended to provide support for a fixed or removable dental prosthesis in the form of a single tooth, partial-arch or full-arch restoration.
- MAX benefits:
- preserving bone and soft tissue
- reduces buccal bone resorption
- limits sinus pneumatization – the longer the implant, the better the prognosis
- faster rehabilitation
- documented minimal bone remodeling
The MAX implant
The 10 Commandments of the MAX® implant

1. Select for purpose
The original intended purpose of the MAX® was to facilitate immediate placement in a molar socket.
2. Never use in thin biotypes
This is generally recommend-ed for all implant types, but is especially important in the MAX®. The socket should be intact in all 4 directions; if not, it is best to augment and delay placement.
3. Never attempt a conventional extraction
This is especially true for molars with divergent roots. Forceps should only be used to assist with initial loosening. The recommended protocol is to decoronize the tooth, separate the roots without removing any interradicular bone, and elevate the fragments.
4. Prepare the site incrementally
Step 1 – If there is a sharp interradicular crest, use a carbide burr to flatten the center to create a wider V-shaped profile, with the surface lying 4-5mm above the socket apex (depth can be confirmed with a probe).
Step 2 – The pilot hole should be offset towards the lingual/palatal, and the mesial.The pilot hole is easier to start accurately with a harpoon drill. The location must be accurate before moving to the next drill as it cannot be revised later in the drilling sequence.
Step 3 – Decoronize and drill through the tooth roots up to 5-6mm depth. Depth must be correct before moving to the next drill as it cannot be revised later in the drilling sequence
Step 4 – Elevate the roots. The recommended drilling sequence after elevation is:
- 2mm twist drill
- 3.3mm or 3.5mm drill
- 4mm spade
- 5mm spade
- 6mm spade or dedicated MAX® drill
- Tap or dedicated MAX final drill
5. Never engage the buccal wall
Filling the socket with the implant should be avoided – there should be a jump gap of 1-2mm. For this reason, 6, 7, and 8mm MAX® have become more popular and 9 and 10mm MAX® are rarely ever used.
6. Place to sufficient depth
Platform of the implant must lie at least 1-2mm beneath the lowest point of the buccal wall. Depth must be pre-planned and established right at the start of site preparation, as subsequent drills are designed only to widen the site and cannot increase the depth.
7. Verify position before placement
This must be done clinically and radiographically. Using a tap to verify placement gives the added advantage of assessing insertion torque.
8. You will encounter a high resistance torque
Mathematically, torque is proportional to the square of the diameter, so increases exponentially as implant gets wider. Therefore high insertion torques with MAX® implants are inevitable and not unsafe. Recommended insertion torque is 70Ncm.
9. Close voids and support soft tissue
Use a wide healing abutment and suture to ensure soft tissue sup-port. If there are particularly large voids, these can be filled with e.g. haemostatic collagen sponge, but leaving the clot is preferred by some and equally acceptable.
10. Follow-up
Post-operative instructions include no rinsing for one week. Follow-up is important not only for the patient but also to contribute to the data and protocol development.
Please visit www.southernimplants.com for more information.
Before deciding rehabilitation through a MAX® procedure, careful pre-operative planning should be done. It is recommended that a pano and intra-oral radiograph is taken. In cases where a clear estimation of available bone can’t be made a CBCT is recommended. Three different types of sockets have been described (Smith and Tarnow, Int J Oral Maxillofac Implants 2013; 28:911-916) and can be used as a guidance to approach the replacement of the molar tooth. Depending on available bone, different surgical approaches are recommended.
Type A
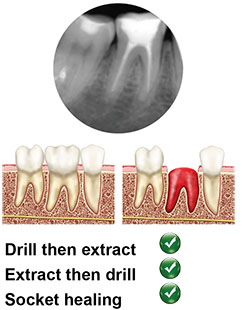
The coronal poriton of the implant is completely contained within the septal bone. Predictable surgical approach either by extracting the tooth and preparing the osteotomy or preparation through the remaining root. High primary stability of the implant is rarely a problem. High torque is normally needed to fully seat the implant.
Type B
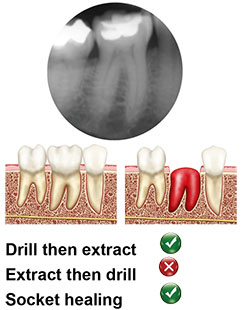
The implants is stabilized but not completely contained by the septal bone; a gap is present between the implant and inner socket walls. In these cases, it is recommended to decoronise the tooth and make the osteotomy though the root to gain as much stability and support from the remaining root as possible.
Type C
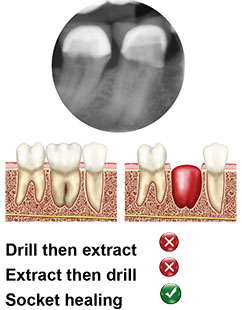
No septal bone is available for implant stabilization. A wide diameter implant must engage the inner aspects of the walls and/or bone apical to the socket to be stable. Consider extraction and conventional 3-4 months soft tissue healing. However, if bone is available apically of the socket and not interfering with the n.mentalis/sinus maxillaris, there are no contra-indications to placing the MAX®.
A word from Graham
Graham Blackbeard, CEO and Founder of Southern Implants

As most readers are probably aware, at Southern we approach most matters with a fair degree of freedom, to encourage innovation and fresh thinking. This will apply to our next Forum, in Hermanus, April 15-18, 2018. The Southern Forums are unscripted, limited in size and arranged to facilitate interaction between esteemed researchers, leading academics and top clinicians. If you are in one of these categories, ensure to register early. Please contact your local office for registration details.
We aim to devise protoocols and products that result in implant restorations that are stable over long periods of time. We gauge our success by analyzing 5, 10, and 20 year outcomes. The keys to long term success are revealed, bit-by-bit, as research is accumulated. Recent studies drove home a clear message: If you reduce the stresses in the system (stress in implant, stress in retaining screw and stress in abutment), the system has greater long term stability … no, or minimal bone loss over time, soft tissue stability, and the reduction or elimination of complications.
Sub-crestal angle correction does exactly that: if you compare the stresses in a Co-Axis versus a tilted regular implant, the Co-Axis reduces stress at the implant surface (where it is in contact with bone), by more than 25%. More significantly it reduces the stress in the prosthetic screw, more than doubling the joint stability. Two studies that point to this are:
- Andrew Tawse-Smith et al have published the 5 year results of the Prospective Co-Axis study, where the implants were placed immediately and loaded immediately … no soft tissue recession over 5 years and minimal bone remodeling (0.5 to 0.8mm).
- Greg Boyes-Varley et al have submitted a manuscript covering the 5-12 year follow-up of 417 Co-axis implants…… two late failures and an overall success rate (Albrektsson Zarb criteria) of 99.5%.


Research highlights
Tamsin Cracknell
Southern Implants is fortunate to have been involved in five high-quality publications so far in 2017. We extend our thanks to these researchers and their teams for choosing Southern Implants products for their patients and investigations.


#site_preparation
Prof Marco Esposito(1) and colleagues at the University of Bologna have published the much anticipated one-year results of their split-mouth RCT comparing piezoelectric site preparation to conventional drilling. The authors made use of Southern Implants oncology fixtures. These zygomatic-like implants have a machined-surface coronal section, indicated specifically for the prevention of biofilm growth and soft tissue infection around unavoidable fenestrations. Feedback from the authors regarding their experience with the implants and instruments resulted in technical improvements on several products, and we extend thanks for their valuable feedback.
1. Esposito M, Barausse C, Balercia A, Pistilli R, Ippolito DR, Felice P. Conventional drills vs piezoelectric surgery preparation for placement of four immediately loaded zygomatic oncology implants in edentulous maxillae: results from 1-year split-mouth randomised controlled trial. Eur J Oral Implantol. 2017;10(2):147-58.
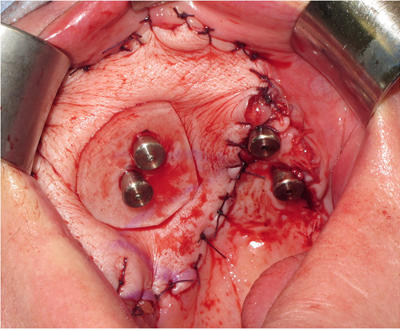
ZIP flap case demonstrating the use of a perforated polythene “washer” to keep the flap from overgrowing the implant abutments during the healing phase.
#oncology
Dr Chris Butterworth and his colleagues at the Department of Oral & Maxillofacial Surgery, University Hospital Aintree in Liverpool(2), continue to break ground in the surgical and prosthetic rehabilitation of maxillectomy patients. Butterworth and Rogers describe the “zygomatic oncology implant perforated micro-vascular soft tissue flap (ZIP flap)” technique, an innovative surgical approach to drastically reduce treatment time for low-level maxillary malignancy. These cases employ Southern Implants zygomatic and oncology implants, together with a fascio-cutaneous flap to cover the defect, enabling early loading in maxillectomy patients. A fixed prosthesis can therefore be provided within weeks of resection.
2. Butterworth CJ, Rogers SN. The zygomatic implant perforated (ZIP) flap: a new technique for combined surgical reconstruction and rapid fixed dental rehabilitation following low-level maxillectomy. Int J Implant Dent. 2017 Dec;3(1):37.
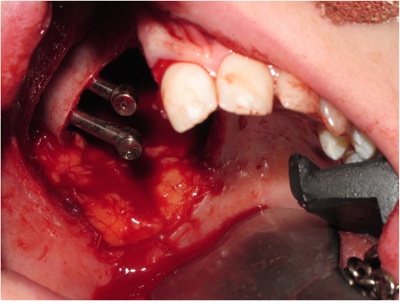
Low-level right-sided maxillectomy with the insertion of two zygomatic oncology implants at time of surgery
#oncology
From the same unit, Dr Dattani et. al. present the first reported use of oncology implants in a paediatric patient(3). The 13-year old male underwent a hemi-maxillectomy due to myxoid spindle cell carcinoma, and received a bar-retained acrylic obturator supported by two Southern Implants oncology fixtures. After two years, the patient is exhibiting excellent function, clinical health, and no adverse effects on facial or dento-alveolar growth.
3. Dattani A, Richardson D, Butterworth CJ. A novel report on the use of an oncology zygomatic implant-retained maxillary obturator in a paediatric patient. Int J Implant Dent. 2017 Dec;3(1):9.
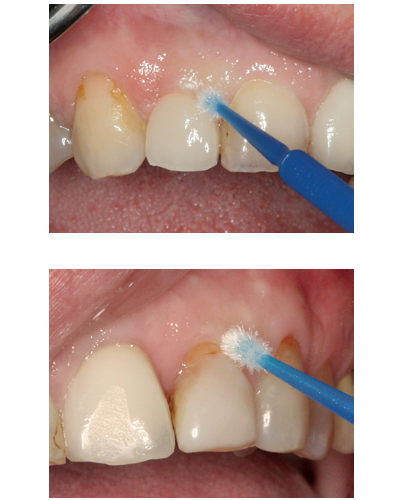
Clinical buccal view of the dental implant (top) and tooth site (bottom) with microbrushes used to collect cytologic peri-implant mucosa smears.
#peri-implantitis
At the Sir John Walsh Research Institute of Otago University, New Zealand, Dr Andrew Tawse-Smith investigated the release of metal particles from titanium-zirconia interface wear in 16 patients at five year recall(4). The study looked at both clinical and cytological outcomes, finding that although peri-implant Ti particles were detected, bone stability and soft tissue health were optimal and unaffected. The patients had received single Southern Implants Co-Axis 12° fixtures, featuring the unique angulated-platform design.
4. Tawse-Smith A, Ma S, Duncan WJ, Gray A, Reid MR, Rich AM. Implications of Wear at the Titanium-Zirconia Implant-Abutment Interface on the Health of Peri-implant Tissues. Int J Oral Maxillofac Implants. 2017 Jun;32(3):599–609.

#co-axis
Prof Dale Howes (Department of Oral Rehabilitation at the University of the Witwatersrand) expands on this topic in the July/August issue of Compendium of Continuing Education in Dentistry(5). In this article, Prof Howes (co-inventor of the Co-Axis) describes the anatomical and prosthodontic challenges that inspired the “subcrestal angular correction” concept – namely, the ability to tilt the implant to make best use of available bone, without sacrificing the occlusal parallelism of the restorative platform. This is an essential read for clinicians seeking to understand the function of the Co-Axis and its multitudinous applications.
5. Howes D. Angled Implant Design to Accommodate Screw-retained Implant-supported Prostheses. Compend Contin Educ Dent Jamesburg NJ 1995. 2017 Jul;38(7):458–463; quiz 464

Head Office and Manufacturing: South Africa
South Africa - Head Office 1 Albert Road, Irene, Centurion, RSA Phone: +27-12-667-1046 | Fax: +27-12-667-1029 | info@southernimplants.com
Australia
Southern Implants Australia Phone: +61-2-8076-9337 Fax: 13002-IMPLANT (46752) info@southernimplants.com.au Terms and Conditions
Benelux
Proscan BVBA Phone: +32-11-822-650 Fax: +32-11-822-651 info@proscan.be
Germany
Southern Implants, Vertriebs GmbH Phone: +49-7121-490-620 Fax: +49-7121-491-717 info@southernimplants.de
Greece
Southern Implants Phone: +30-210-898-2817 Fax: +30-210-898-2232 info@southernimplants.gr
Italy
Southern Implants / S.I.R srl Phone: +39-045-568-351 Fax: +39-346-227-1165 info@southernimplants.it
Namibia
Skydancer Phone: +264-61-235-630 Fax: +264-88-613-840 implants@skydancer-cc.com
New Zealand
Ostralos Phone: 0800-2-46752 Fax: 0800-2-IMPLANT sales@ostralos.com
Portugal and Spain
Contactodent Phone: +351-214-693-332 +Fax: 351-214-693-329 southernimplants@sapo.pt
Russia
Southern Implants Russia/ SIR LTD Phone: +7-926-659-23-35 Fax: +7-965-178-00-69 info@southernimplants.ru
Scandinavia
DAB Dental AB Phone: +46-8-506-505-48 Fax: +46-8-506-505-00 kontakt@dabdental.se
Turkey
Bioport Co. Phone: +90-212-272-7577 Fax: +90-212-272-7628 info@bioport.com.tr
UAE
Southern Implants JLT Phone: +971-50-857-0357 Fax: +971-4-427-5011 info@southernimplants.net
United Kingdom
Southern Implants UK Phone: +44 20-8899-6845 / 6 / 7 Fax: +44 20-8899-6843 info@southernimplants.co.uk
USA and Canada
Southern Implants North America Inc. Phone: +1-561-472-0990 Fax: +1 561-472-8401 customercare@southernimplants.com


